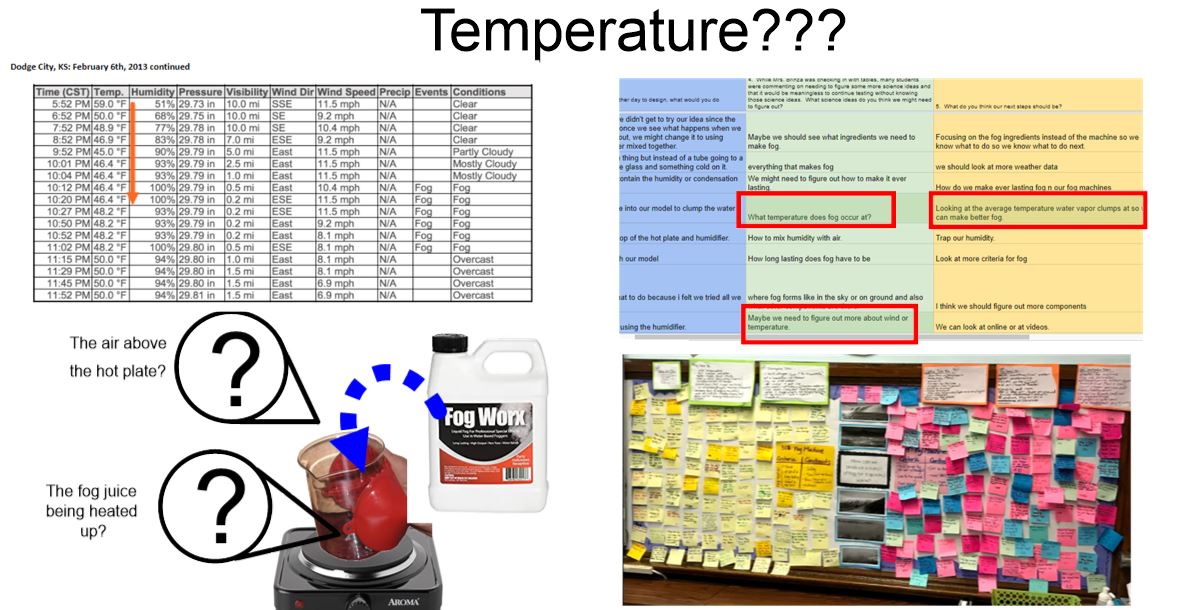
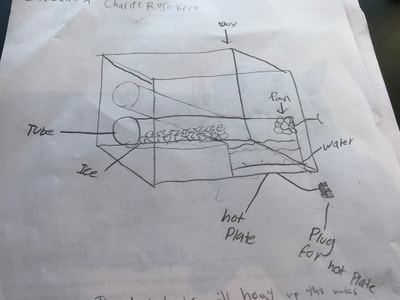
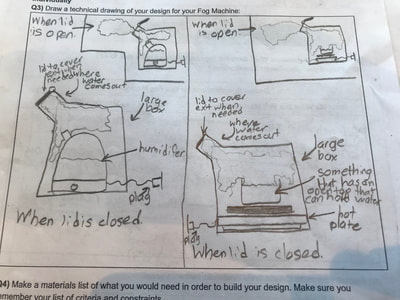
So we figured out a lot about water and what happens to it as it is heated and cooled. Since we know fog has something to do with water, this makes sense. But it also makes sense to investigate what other other substances do with hotter and colder temperatures since other things like CCNs and air play a role in making fog. So overnight, students brainstormed some Investigation Ideas to test this in Google Forms. Here are some of their results, and after a discussion...
We began the investigations, noticing what happened, and developing some models to see if air and other substances behave like water when it is heated and cooled.
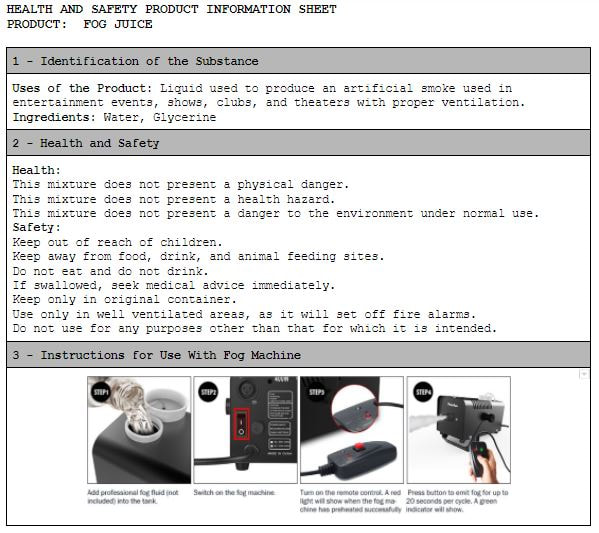
| | |
Heating Up A Bottle with Bubble Solution on Top | Heating Up a Bottle with a Balloon on Top | Heating and Cooling Vinegar (Using pH Paper as an Indicator) |
| | | |
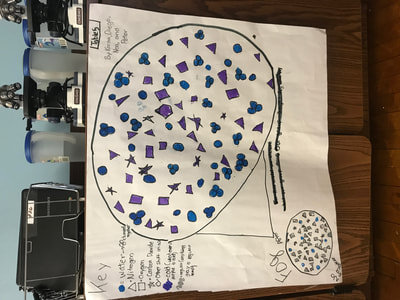
We figured out a lot from all these investigations, and it seems that water, vinegar and air molecules all act the same way:
1. Molecules move faster and take up more space when they're heated.
2. Molecules move slower and take up more space when they're cooled.
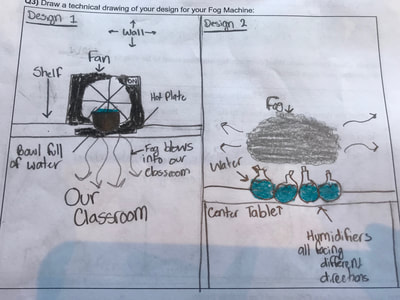
From all our investigations, though, we would LOVE to see what is really going on with the molecules, since our ability to show movement with them is kind of limited being static on paper. Is there a way we can better see what's going on at the molecular level...IN REAL TIME?
1. Molecules move faster and take up more space when they're heated.
2. Molecules move slower and take up more space when they're cooled.
From all our investigations, though, we would LOVE to see what is really going on with the molecules, since our ability to show movement with them is kind of limited being static on paper. Is there a way we can better see what's going on at the molecular level...IN REAL TIME?










































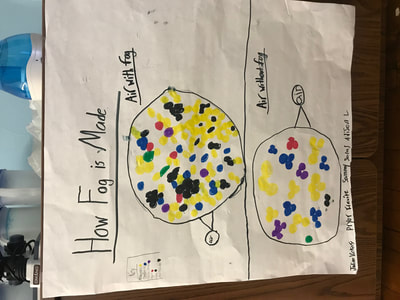













 RSS Feed
RSS Feed