Sixth graders are visiting their DQB one last time to answer as many of the questions that they set out to answer. And BOY, was it a lot! Way to go 6th grade...you are FOGSTARS!
|
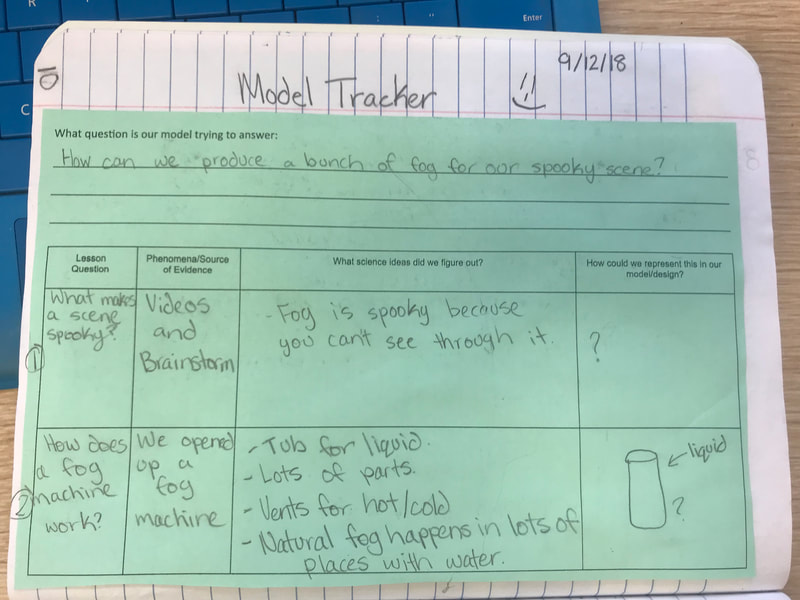
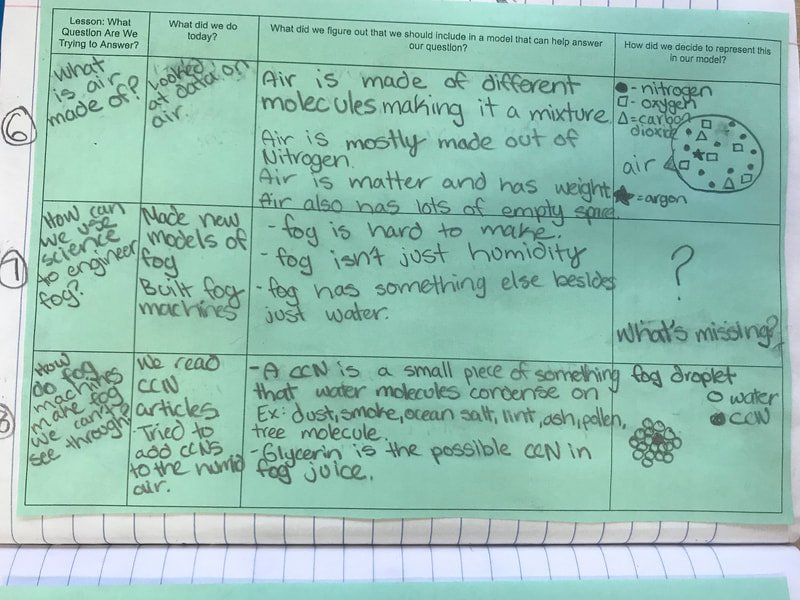
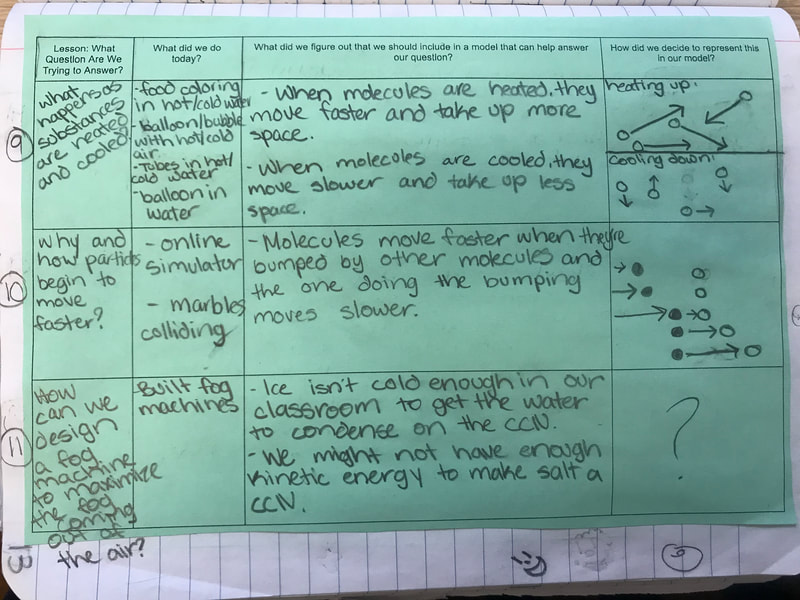
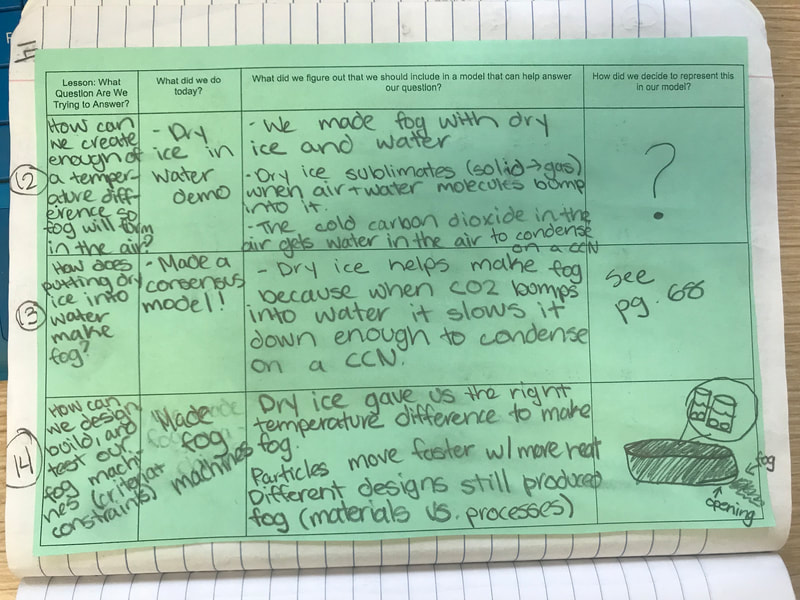
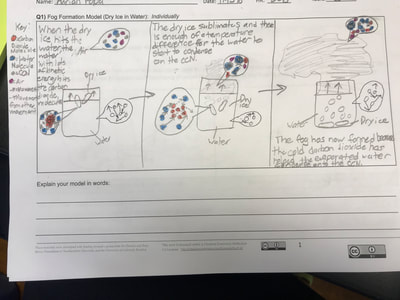
All throughout our unit, we've been documenting what we've figured out in our Model Tracker, a handle tool that has helped us summarize our learning. Check out a student's work from one class here, and now that our Fog Unit is nearly done, you can see how we put lots of pieces together over time!
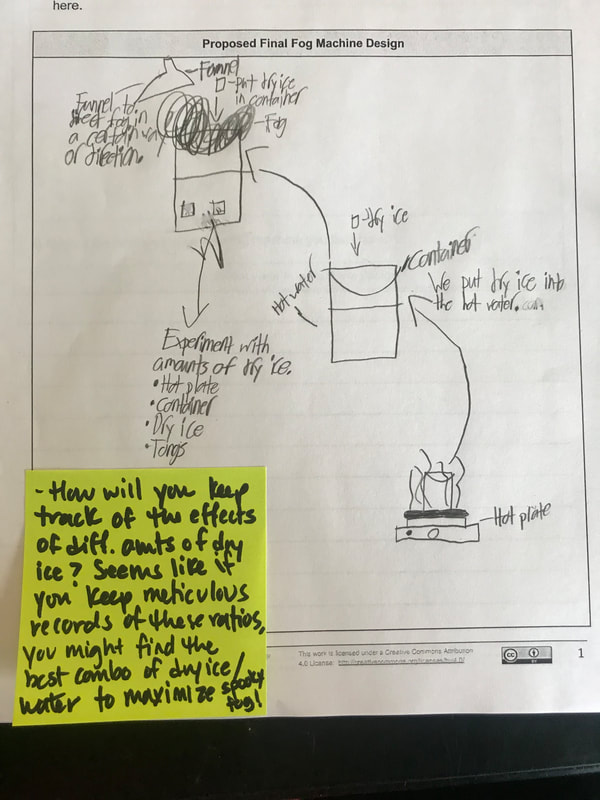
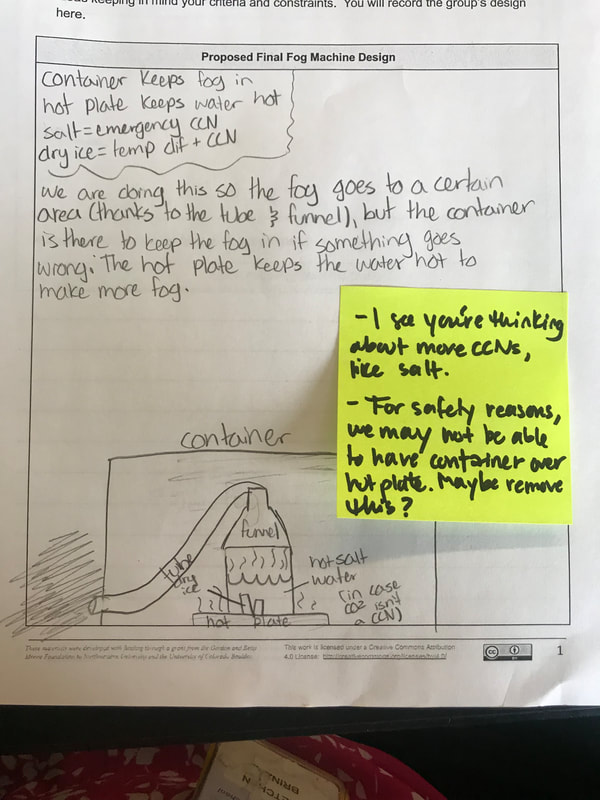
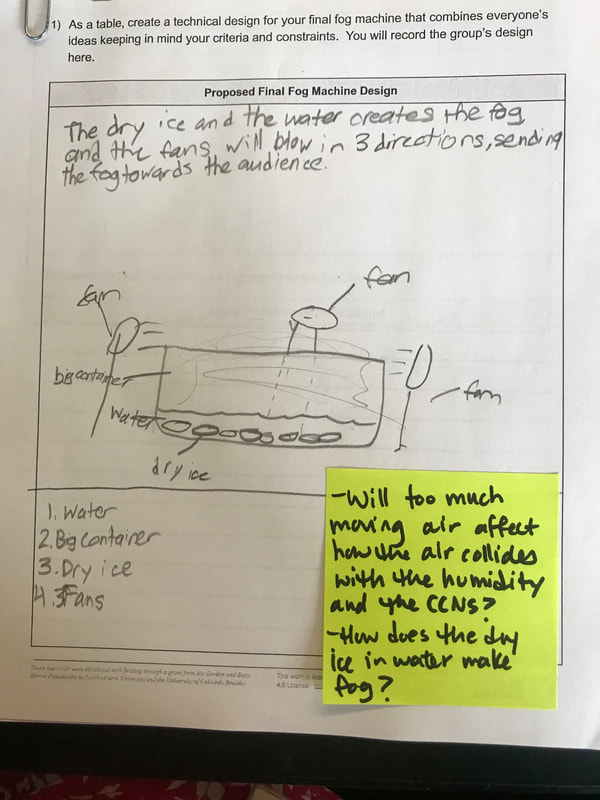
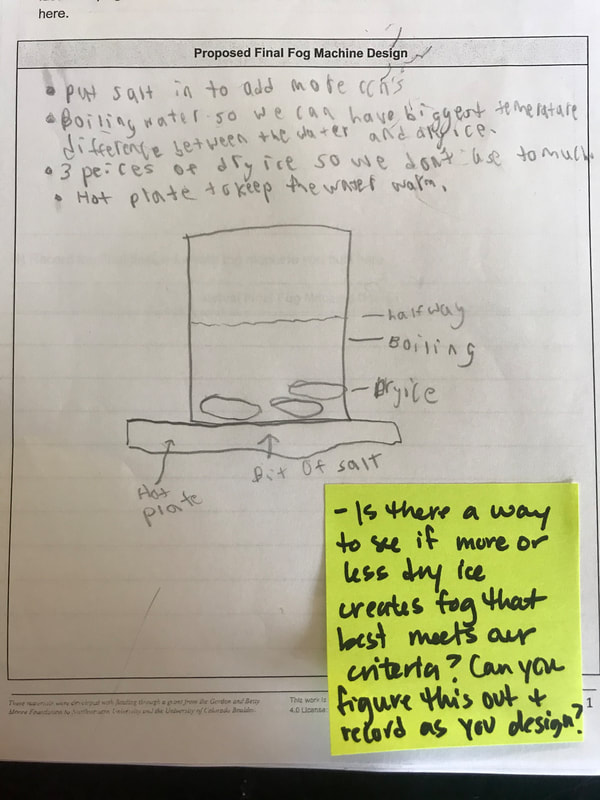
We had such a blast presenting our final fog machine designs! With different ratios of dry ice: water, to different CCNS (salt, CO2 or spray deoderant), to different temperatures of water, to even different ways to dispense of the fog, we can certainly cay we made spooky fog!
While we feel successful with our fog machines, we still have some more designing to do to make our classroom super spooky (this will emerge as the year continues and we get ready for our STEM Fair in the spring). We also want to connect all that we learned back to the cities' data that helped us figure out the conditions/factors that enable fog to form. We'll be looking at city-specific factors as we wrap up our fog unit early next week! Now that we understand the science behind fog formation, we're on to putting these science ideas into place by using the engineering design process! We've got two days of testing before we present our final ideas to the class to see who can best meet our criteria and not violate our constraints!
Students signed safety contracts today and began testing...we've got some interesting ideas on the table, and some spooky fog is in the works! We did it folks! We finally came to consensus today putting together all our science ideas behind what makes fog, and we did it with dry ice and water!
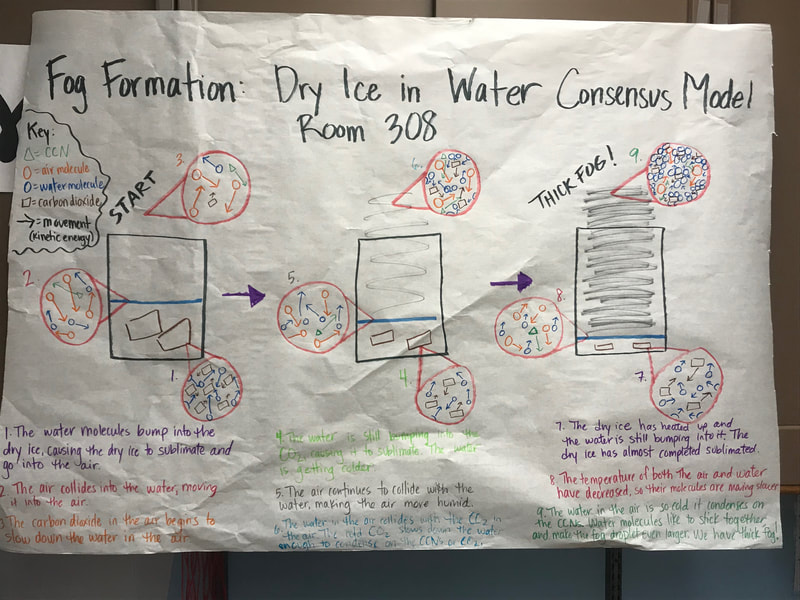
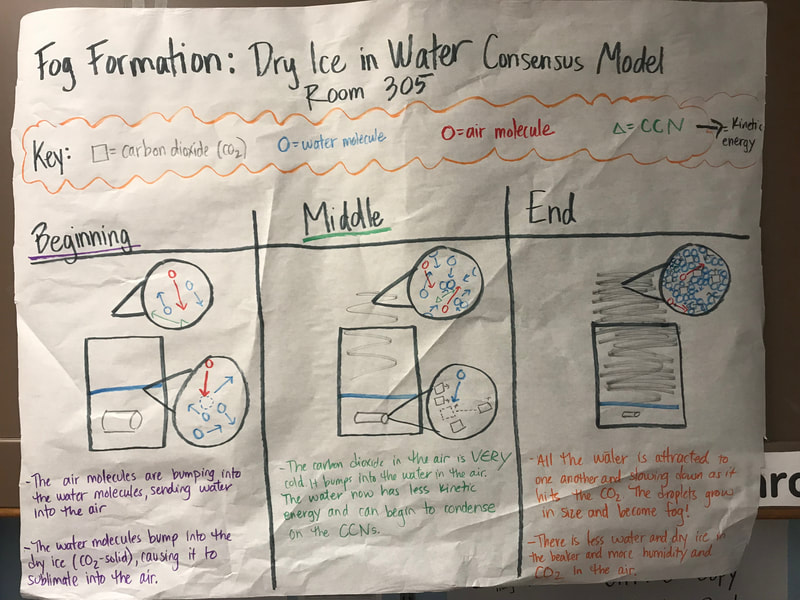
To make a long story short, we figured out the following: 1. When you put dry ice in water, the water molecules have more kinetic energy than the dry ice molecules. This means that when they collide into one another, the energy from the water transfers to the dry ice (solid CO2). This is what gets the dry ice to sublimate (turn from a solid to a gas) and put CO2 into the air. 2. The water in the beaker gets into the air because the air above it is colliding into it, getting it to evaporate. This is making the air more humid. And we know that high humidity is a necessity for fog formation! 3. Once the CO2 and water are in the air, they do two things. First off, we figured out that water molecules are attracted to one another. And since they are still moving, when they bump into the CO2 molecules (which are STILL REALLY, REALLY COLD), they slow down the water molecules enough to get them to condense on either nearby CCNs or the CO2 (which we're thinking is a CCN, too). This high humidity in the air, along with the cooling (because of the dry ice), allows fog droplets to form and fog to appear! Students are now thinking about their fog machine designs that can best meet the criteria and constraints! Working in teams, they submitted designs to Mrs. Brinza while she secures dry ice. Check out some of the groups' design ideas and some of the feedback Mrs. Brinza gave them. After looking at students' models last night, it was important for the class to see how each part of the dry ice/water investigation plays a role in forming fog. The class spent time on summarizing what we've figured out so far regarding fog droplets along with the specific roles water, air, and dry ice play. Here are our findings! We're going to take all these ideas, along with the ideas that students shared individually with Mrs. Brinza into account as we establish consensus for our dry ice and water form fog!
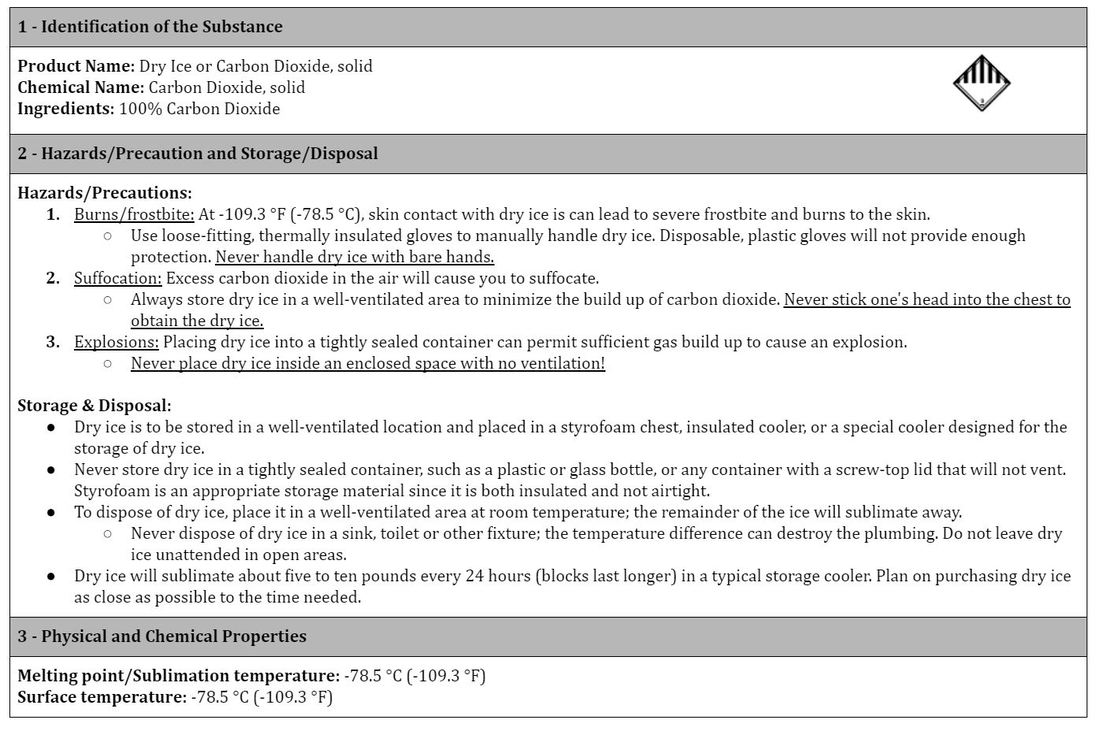
With students suggesting we couldn't get the water in the air cold enough to condense on a CCN, we went the dry ice route. We've figured out from our dry ice safety sheet that dry ice is REALLLLLLLLLLLLY cold, and this would hopefully give us the temperature differential we needed. And sure enough it did!
We're now thinking about how this scientific phenomenon works, knowing it's producing the fog we've so longed for. It's important to understand the science behind something before we try to engineer, and so we're spending some serious time on what this process looks like at the molecular level before we build our fog machines! Check out some sixth graders' models for how fog forms using dry ice and water. We've got some great ideas out there before we establish consensus!
We did all kinds of things with the dry ice and water, including just putting it on the table to sublimate (change from a solid to a gas), along with putting it in water. We had lots of discussion around what the molecules of the dry ice, water, and air were doing alone and with respect to one another. There was lots of talk about collisions and what each molecule was doing to another molecule depending on how much kinetic energy it had. We also used BtB as an indicator for carbon dioxide (dry ice is solid CO2 in case you didn't know), so we were able to tell when it was in the air and in the water.
Lots of students wanted to heat the water, as they know that heating it gets it to move faster, and make more of an impact with the collisions. So we tried that, too, and boy, did we see a difference. While this was all great to witness, before we move forward with our designs we're going to have to really understanding how dry ice and water makes fog. We know the water gets us the humidity part of fog. We know there are CCNs in the air. And we know that dry ice is cold, making the temperature difference we need to slow down the water enough to condense. But is there a way to use all stuff we figured out to make fog even thicker, spookier and longer-lasting than the demonstrations today? Check back soon for our models to understand this process... Out of the last two investigation ideas to make fog (dry ice or liquid nitrogen), Mrs. Brinza went with dry ice first. There's been some talk about using dry ice intermittently throughout our study, so she went with that. Both dry ice and liquid nitrogen are cold, and we're thinking that they might be cold enough to get the humidity in the air to condense on the CCNs that we know are in the air.
So looking at a dry ice safety sheet, students had to convince Mrs. Brinza on whether we should use dry ice, and the overwhelming vote was "NO." Students ideas: 1. Dry ice is TOO cold. We're going to get frostbite/burns and safety is important to reach our constraints. We've got goggles to protect our eyes but we don't have insulated safety gloves. 2. We don't have the right storage container. It's going to blow up the cooler Mrs. Brinza has because it's sealed tight, and as the dry ice sublimates, we don't want an explosion in our classroom. 3. Our classroom isn't well ventilated, and we don't want to suffocate and die. 4. It's expensive to get and ship. Dry ice is anywhere from $1-3/pound, but the shipment and delivery charges ADD up quickly, not meeting our "affordable" constraint. SO... We talked about using dry ice to be our last-ditch effort at making fog (or liquid nitrogen). We walked through each of their concerns about safety and cost, and came up with the following: 1. Mrs. Brinza will be the one to touch the dry ice. She's committed to getting low-cost insulated gloves this weekend to protect her skin. Everyone will wear goggles to protect their eyes. 2. When getting regular ice at Mariano's grocery store this week, Mrs. Brinza saw loose-lid Styrofoam coolers that were around $6. That's fairly low-cost. And it will allow the sublimating dry ice to escape and not build up pressure in the cooler. 3. We will open the windows and blow fans to keep the air moving in our classroom so our room is well-ventilated and no one suffocates. 4. Mrs. Brinza will attempt to get a small amount of dry ice. We'll do some things with it that we think will cause fog, and if it works, we will figure out exactly how to use it in our designs before we get a BIG shipment of it. Heck, it might not even work, so getting a small amount will lie within our constraints of cost. 5. We don't want anyone to get hurt, so we will keep the Styrofoam container of dry ice in a safe, out-of-the-way place, clearly identified with a safety set-up (like a sign that screams "DO NOT TOUCH!"). Kind of like those "FLOOR IS WET" signs when someone spills something that has been mopped up. We are hoping that Mrs. Brinza can get some dry ice over the long weekend (thank you, Veterans). Fingers crossed! So we really thought we were going to make fog today. And we failed. Yet again.
We had the heating of the water and CCN mixture to get them into the air. We had ice to condense the water onto the CCNs. But why weren't we getting fog? We are thinking we weren't able to make it cold enough. We had TONS of ice. But if the gaseous water molecules in the air were colliding into the solid water molecules in the ice, the water molecules in the ice might not make the humidity slow down enough to condense. So what are our options then? We went back to our original Investigation Ideas Poster we created at the start of the unit when we agreed to make spooky fog, crossing off all the investigations we've done. And the only ones left involve using dry ice and liquid nitrogen, neither of which is apparently safe. But they are cold, and can possibly help slow down the humidity in the air enough to condense on a CCN. Can the class convince Mrs. Brinza to use either of them? |
Driving QuestionHow can we produce a bunch of fog for a spooky scene? Archives |











































































 RSS Feed
RSS Feed