So now that we feel we've figured out fog, we're putting all our science ideas into our engineering designs. We've got higher humidity planned out, getting CCNs into the air planned out, and a way for the water in the air to condense on a CCN! Fog soon!!!!!
|
We figured out a lot from all our temperature investigations, and we went back to the DQB to see what we could answer! And boy, could we answer a lot! After figuring out so much, we are in unanimous agreement that we know how fog is formed:
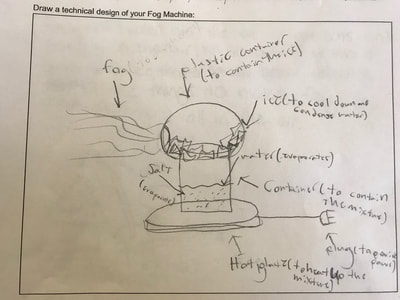
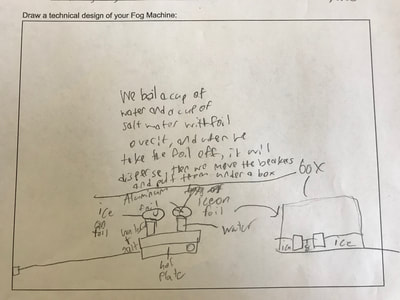
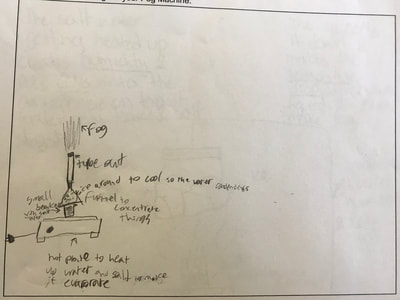
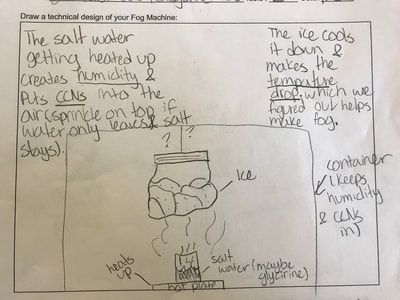
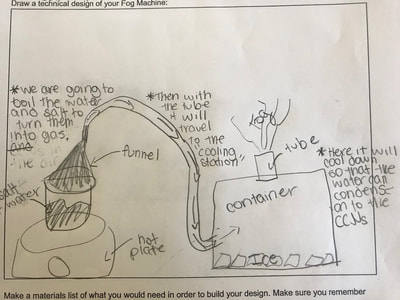
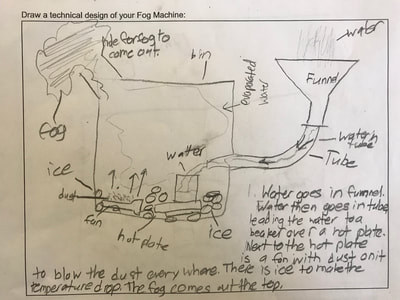
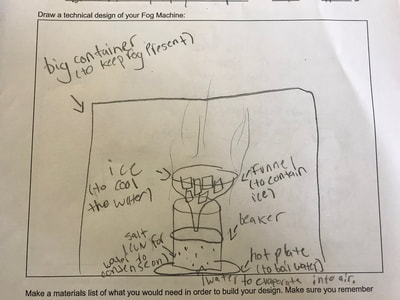
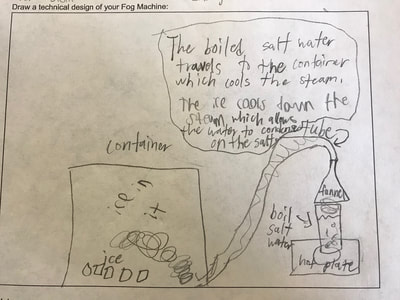
1. Putting humidity in the air by heating up the water molecules. 2. Making sure there are CCNs small enough into the air by heating them up (guaranteeing there are some in the air beyond what are naturally already there). 3. Cooling down the water in the air so it condenses on the CCNs! This all makes sense for the places that have natural fog. They're mostly by a body of water, early in the morning when it's cooler but the sun comes out to heat stuff up! We'll spend a day designing and a day building our fog machines. There have been some requests for glycerine (the obvious CCN in fog juice, which is a mixutre of glycerine and water) and salt (the obvious CCN for fog over the ocean) along with ice...lots and lots of ice!!! So while we feel we know more about temperature now from our air and water investigations, we all agreed it would be better to really know if what we were seeing on a large-scale with our eyes was the same as what was going on at the molecular level. Students wanted a microscope to zoom-in on the molecules. Sadly, our microscopes at school don't zoom in enough. But Mrs. Brinza found an online simulator that we could explore to see what is really going on and how figuring things out could help us better understand how fog forms. From using the simulator below, we found: 1. All molecules do move, regardless of whether a substance is in a solid, liquid, or gaseous state of matter. 2. The more heat is applied to a substance, the more the molecules of a substance move, and the more space they take up. 3. Conversely, the more heat that is taken away from a substance (or cooled), the less the molecules move, and the less space they take up. 4. At one temperature, all the molecules of a substance don't move the same exact speed. This is because as molecules collide into one another, the molecule doing the bumping slows down and its energy is transferred to another molecule, making it move faster. 5. Not every substance is at the same state of matter at the same temperature. For example, water and oxygen are liquid and gas, respectively, at 20 degrees Celsius. We really wanted to watch just TWO molecules, as we are trying to figure out how air molecules, CCNs and water molecules may all be interacting with each other during fog formation. We decided to use marbles, as they are easy to move (and Mrs. Brinza had a whole tub of them--ping pong balls were suggested, but she doesn't have those)! We really saw what was happening between the marbles here, and how this can relate to moving molecules, like liquid water into the air, or CCNs into the air. We know that if we heat a substance, the molecules move faster, and this causes more collisions between molecules. We also realize that after a collision, we will end up with a molecule that slows down and a molecule that speeds up.
With all this knew stuff we figured out, it was time to go back to the DQB to see if we could answer any of our questions! |
Driving QuestionHow can we produce a bunch of fog for a spooky scene? Archives |












 RSS Feed
RSS Feed