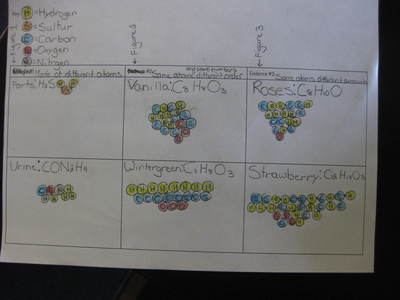
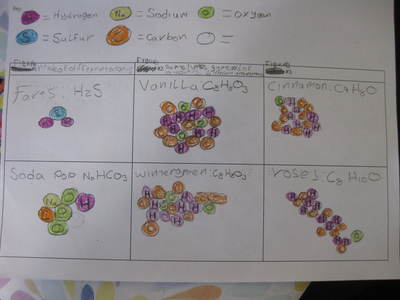
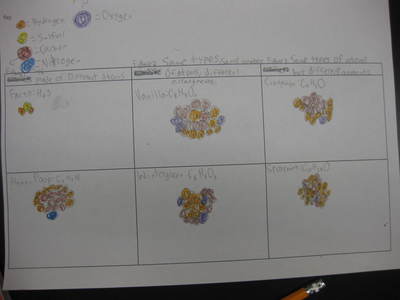
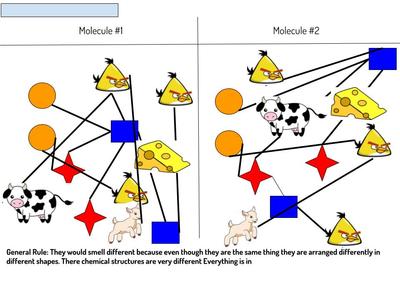
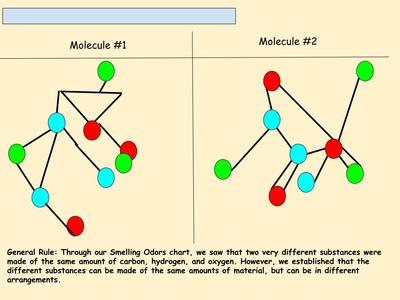
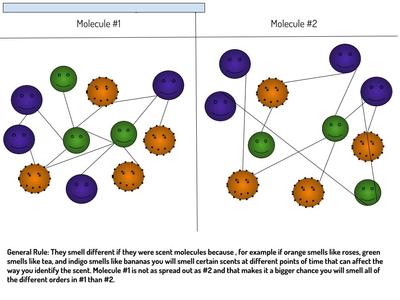
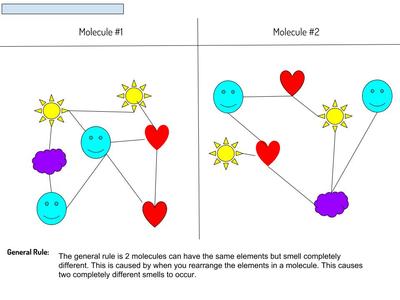
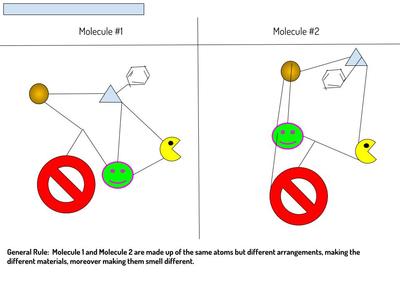
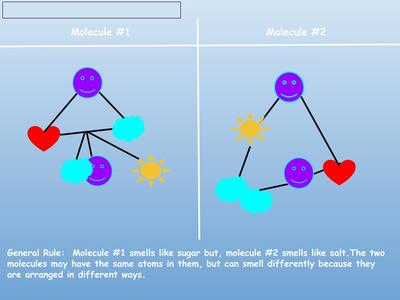
Now that we've figured out how phase changes play a role in smelling things from a distance, we're seeing how understanding the particulate nature of matter is important in some real-world situations. Here are the scenarios that students had to analyze...who knew we'd see (and smell) science everywhere!?!?! (wink, wink!)
|
|












 RSS Feed
RSS Feed