Here is the criteria for their project:
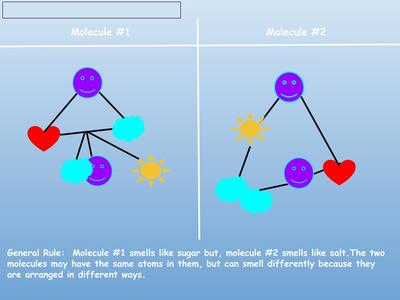
PSA Commercial: How Can I Smell Things From a Distance?
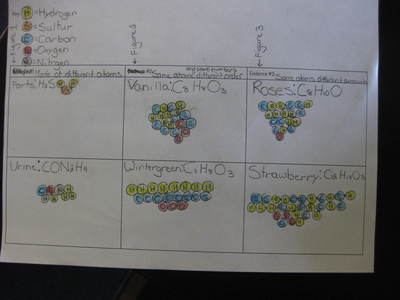
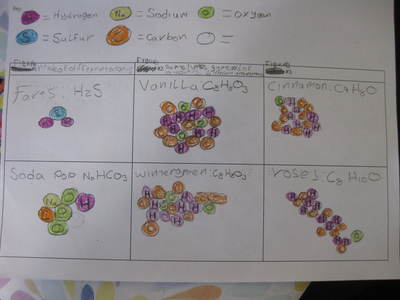
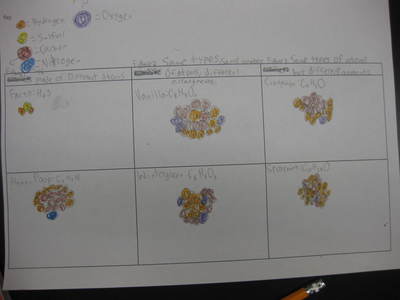
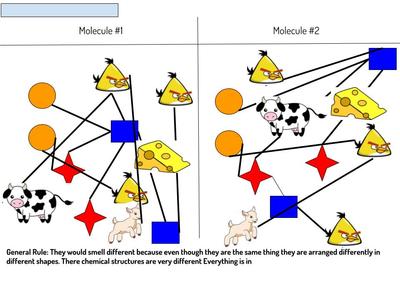
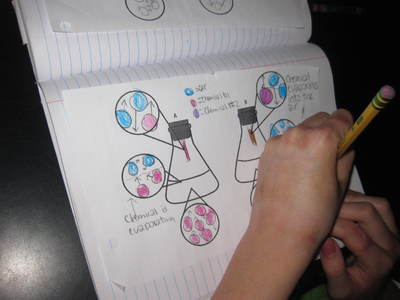
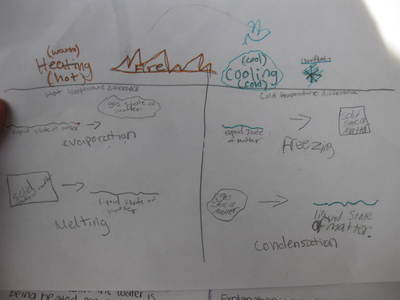
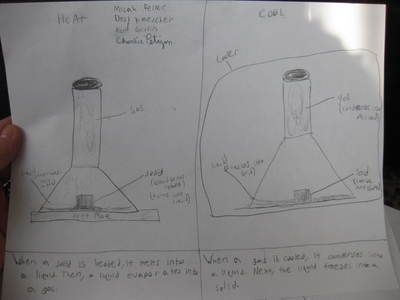
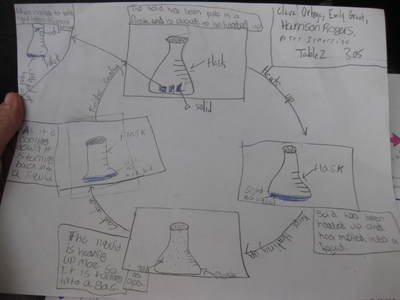
Name: ______________________________________________________________________ Date: _______ Table: __________ HR: ____________ Public Service Announcement: How Can I Smell Things From a Distance? Directions: Your table group will be creating and producing a public service announcement that educ...


































 RSS Feed
RSS Feed