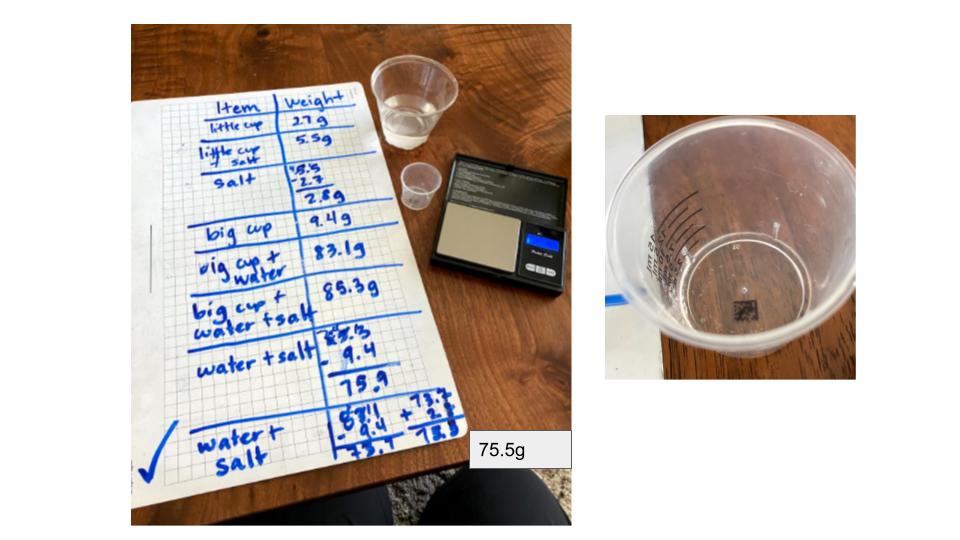
With all this discussion around particles and dissolving, we're starting to see some patterns! We've noticed that our dirty water looks so dirty because there are probably LOTS of solids that have dissolved in them! We continued to focus this week on what it means for a substance to dissolve, practicing with salt, sugar, and a favorite this time of year--citric acid (it's found on the outside of Sour-Patch-Kids). We tasted samples of water with salt dissolved in it and even used a scale to prove that the salt was still there!
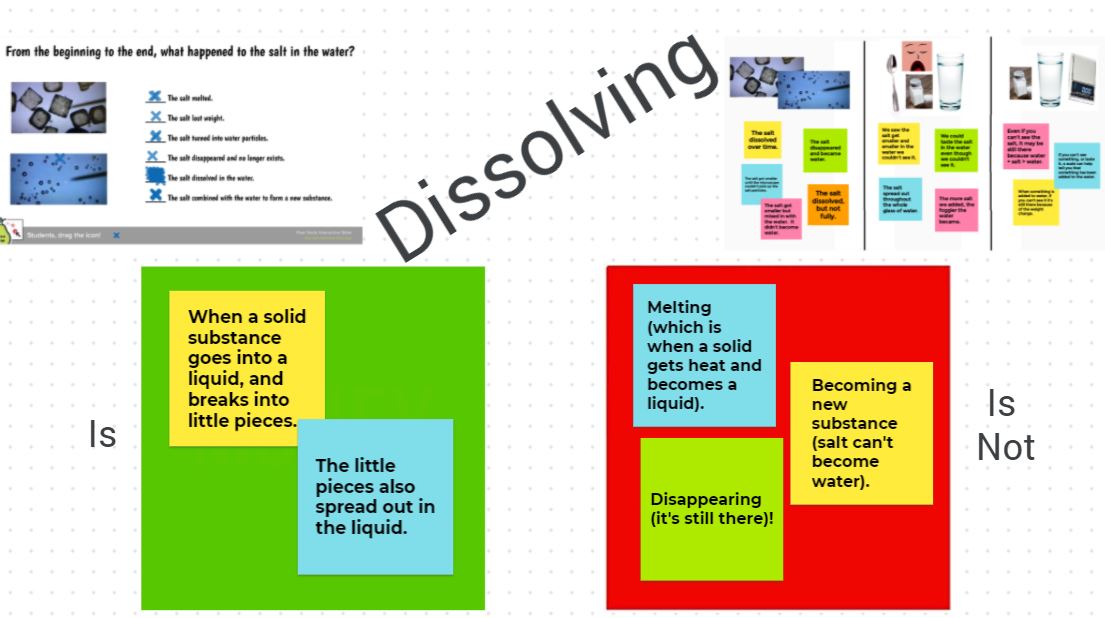
We also came to a consensus on what it really means for something to dissolve in water:
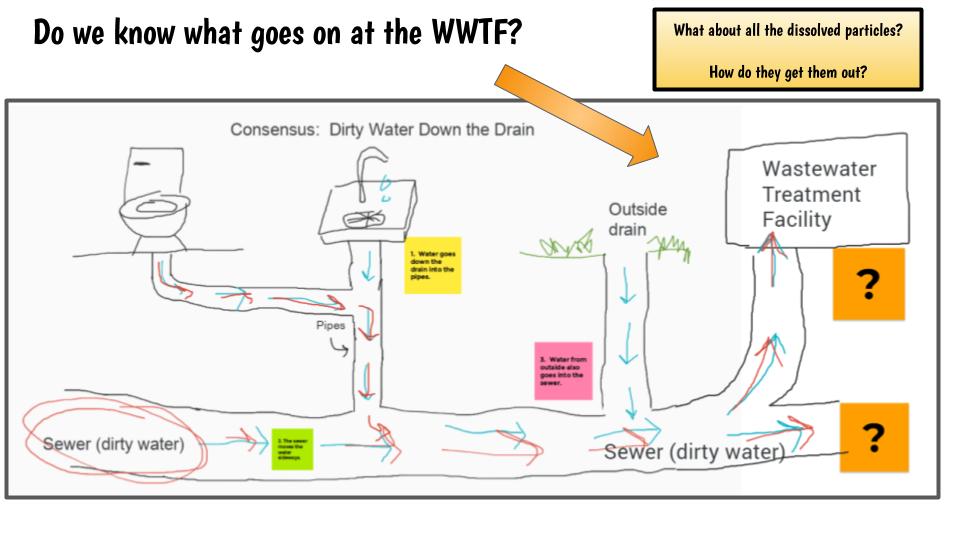
With all this focus on dissolved solids, we're going back to where we had a lot of questions--what exactly happens at the wastewater treatment facility to get out everything from the dirty water? If there are so many tiny, tiny particles in the water, how exactly do they get them out?
 RSS Feed
RSS Feed