We recognized through Dalton's work that we could get a gas by heating water. We didn't think that there would be a new substance, but we also saw that we needed to gather evidence to prove this true!
We saw that heating water certainly produced bubbles, and the gas that was escaping a connected tube also was turning back into a clear substance. We wanted to see what would happen in the flammability test, and this gas extinguished a flame. We realized we couldn't test the density of the clear liquid when it was still hot, so we waited for it to cool down to room temperature.
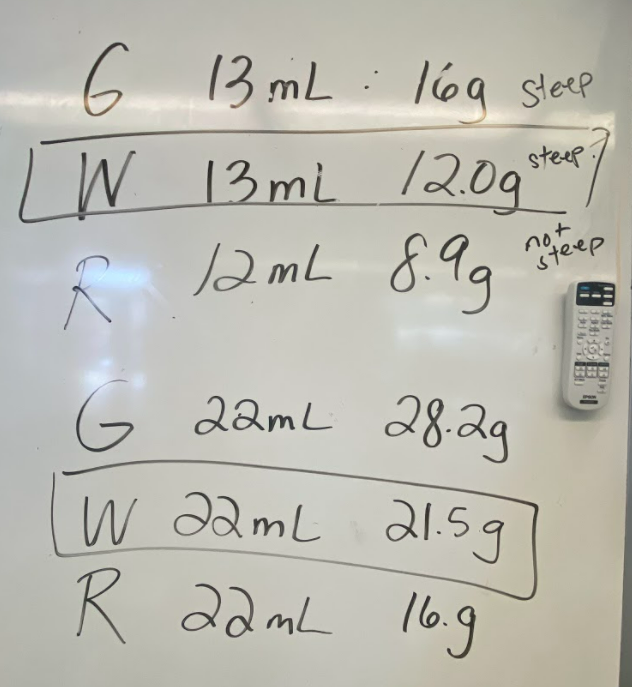
While we waited for the liquid to cool down, we realized we had limited understanding of what density actually was. Looking at the unit for density, we realized it was a comparison of g/mL, which meant it was a unit rate comparing the mass of a substance to its volume. We also began to see that water wasn't the only clear liquid around (duh!) and that the density of a substance is a great way to identify a substance that looks exactly like another one!
While we waited for the liquid to cool down, we realized we had limited understanding of what density actually was. Looking at the unit for density, we realized it was a comparison of g/mL, which meant it was a unit rate comparing the mass of a substance to its volume. We also began to see that water wasn't the only clear liquid around (duh!) and that the density of a substance is a great way to identify a substance that looks exactly like another one!
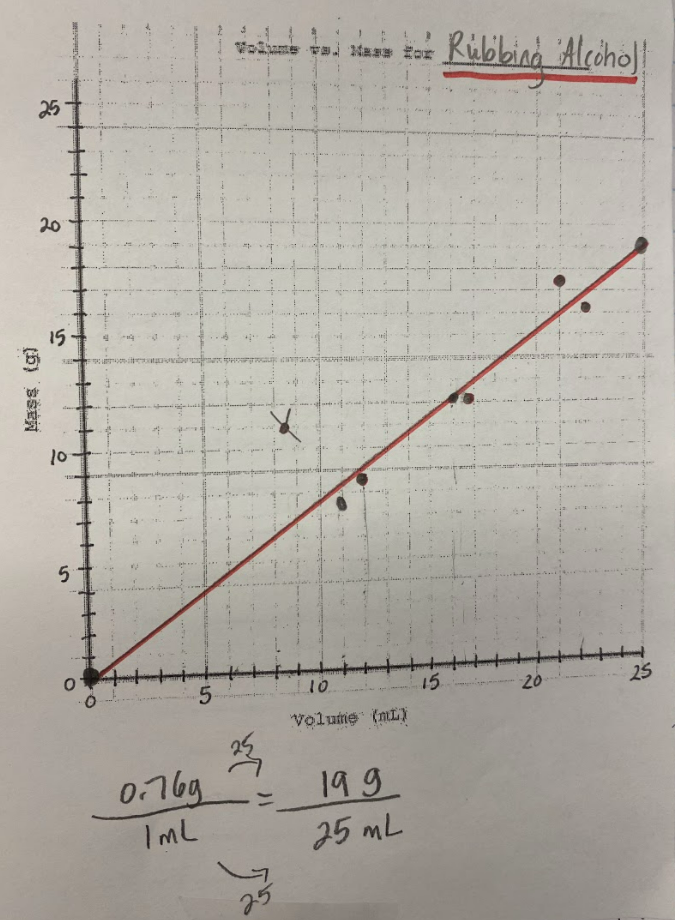
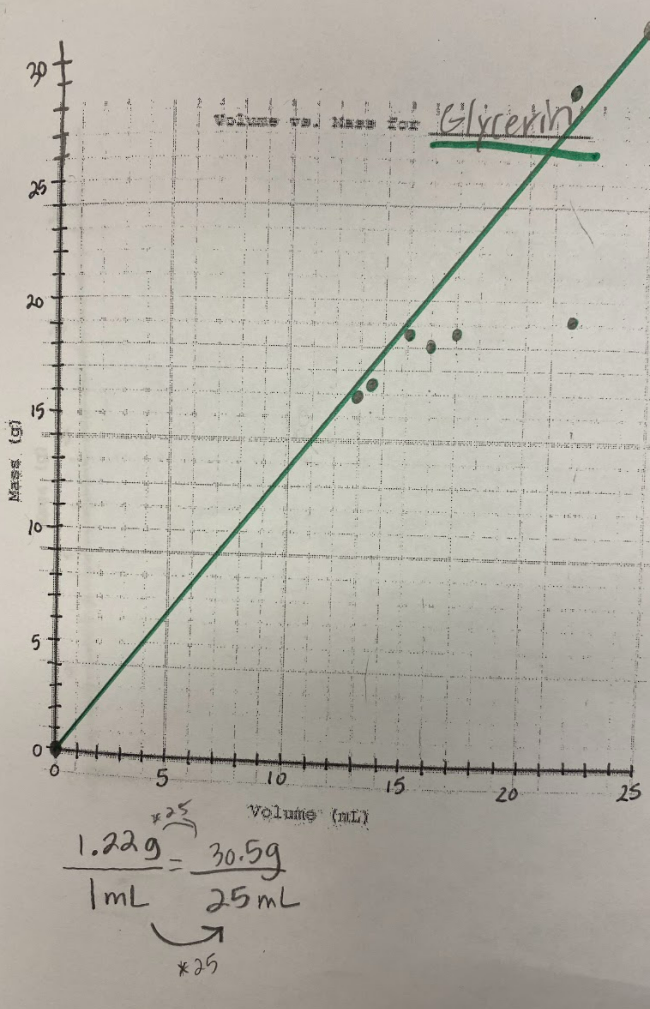
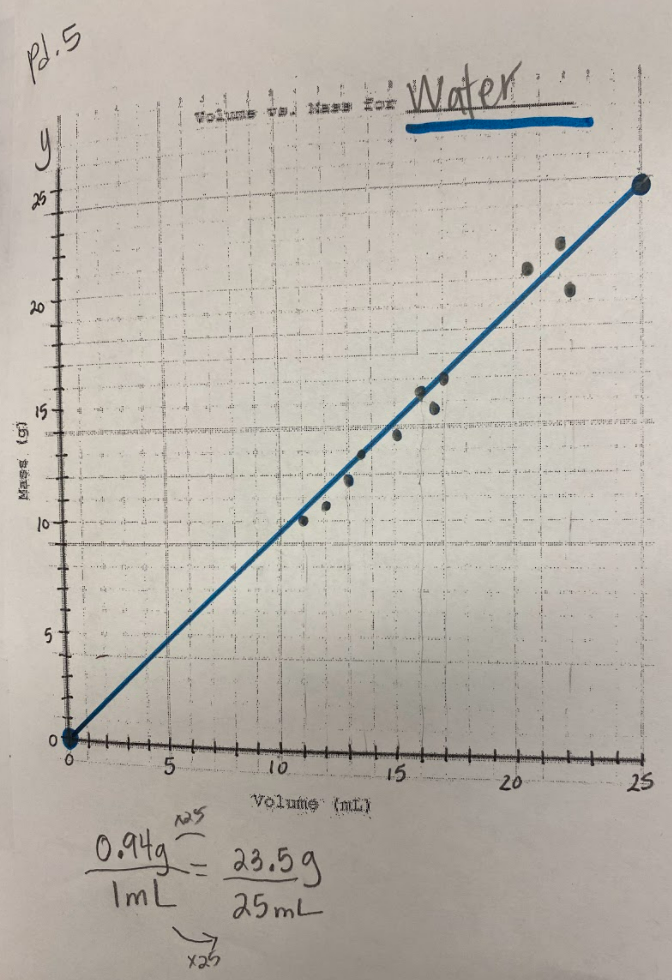
Our efforts to see patterns in the mass vs. volume relationship began to emerge, and we saw that there were diagonal lines getting taller appearing, but at different rates. Rubbing alcohol's density was the lowest, with the least steep line. Glycerin's line was the steepest, with water's line falling in the middle! We also agreed that density was a unit rate, figuring out how much a substance's mass was PER milliliter of volume!
By this time our clear liquid had cooled, and by weighing it and calculating it's volume, we determined the clear liquid's density to be 0.96g/mL. Since we calculated the density of water (when we knew it was water) to be close to this value, we agreed that heating water doesn't produce a new substance, and it's still made of water particles!
So to summarize, we don't end up with a new substance when we heat it! Our bath bomb gas is a new substance, so these processes are NOT the same! Onto running electricity through water!
So to summarize, we don't end up with a new substance when we heat it! Our bath bomb gas is a new substance, so these processes are NOT the same! Onto running electricity through water!





 RSS Feed
RSS Feed