It's always a great day when we summarize what we've figured out by answering all the questions we asked to launch our unit and at other points during our unit!
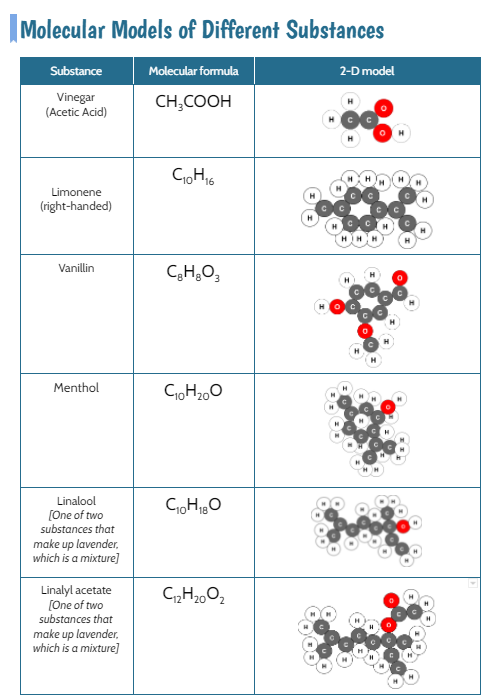
We also are wondering how the odors in bath bombs work. Like what are they? How do they reach our nose? We explored various scents, trying to guess them along with analyzing their molecular structure! We are starting to see some patterns, and with some further research, we came to the following agreements:
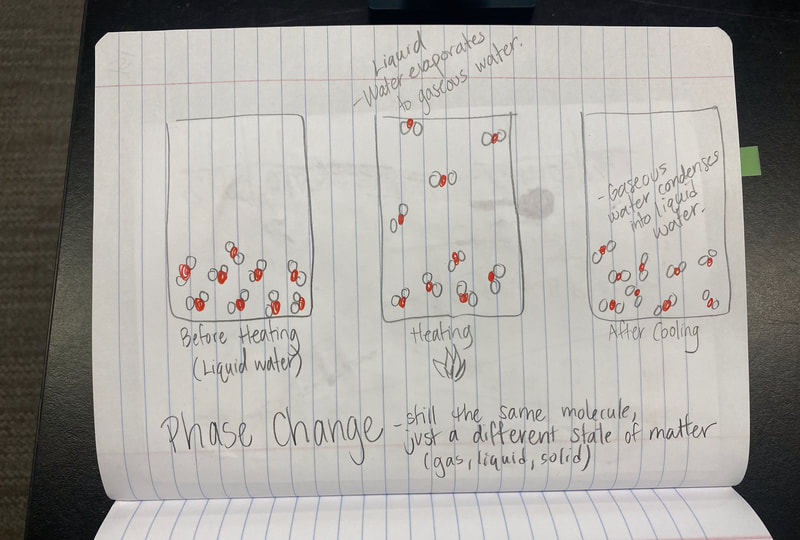
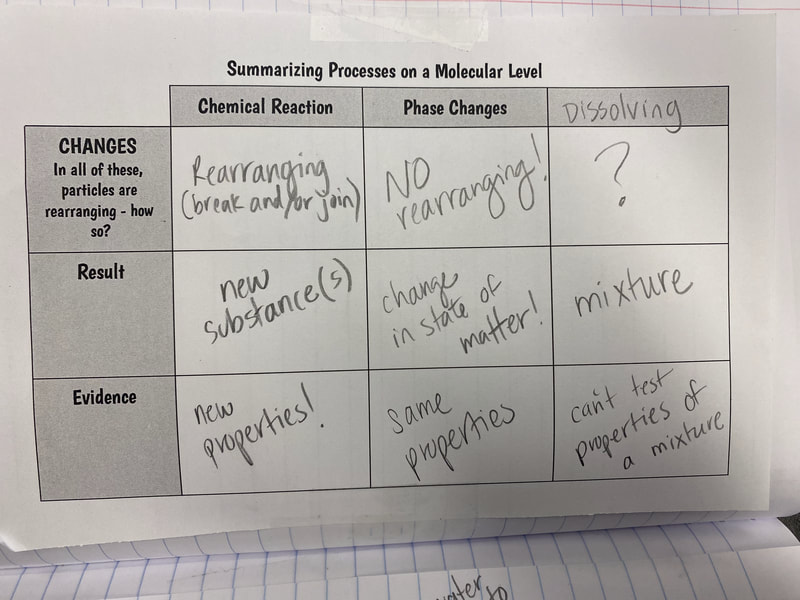
1. Odors are evaporated molecules that reach our nose, and receptors interpret the molecules with our brain's help! 2. Every odor is a different molecule, so odor must be a property. 3. All odors are comprised of different amounts of arrangements of carbon, hydrogen, and oxygen. 4. If one atom from an odor molecule is dropped or added, we end up with a new molecule, and therefore a different substance and odor. That explains why perfectly cooked popcorn smells different than burnt popcorn! Next steps, return to our DQB to answer all the questions we set out to ask when we launched our unit! So if not everything creates a new substance like in a chemical reaction, what happens in other processes like when water boils or when salt dissolves? If these don't make new substances, what happens? We used water molecules as an example to compare the difference between different types of processes we've seen happen! We'll be working on some final unit wrap-ups next week, as we've still got questions on our DQB about other substances in the bath bomb and how some bath bombs smell and others don't!
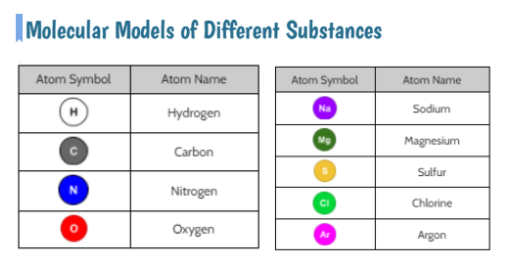
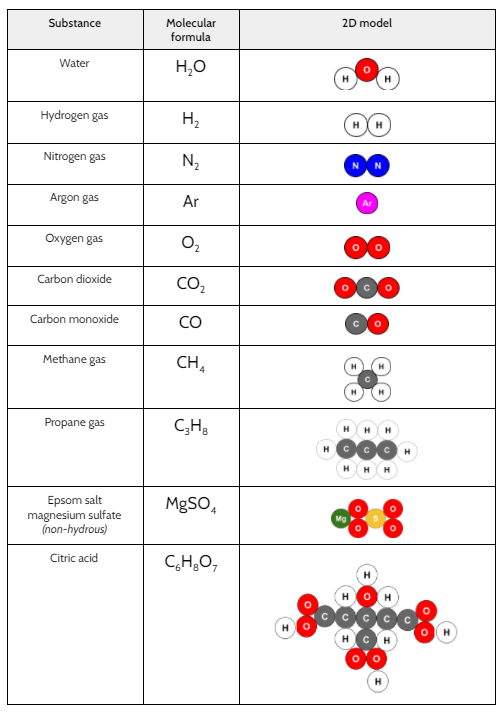
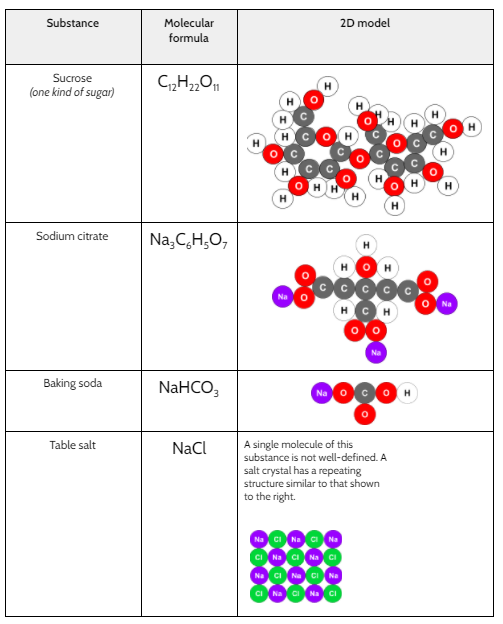
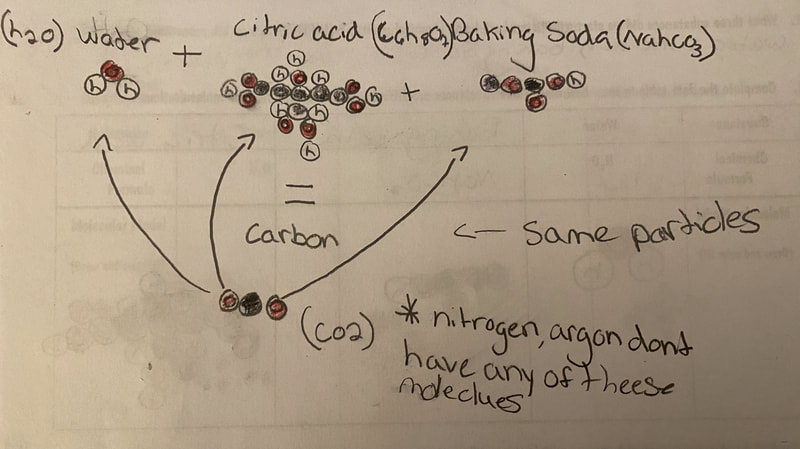
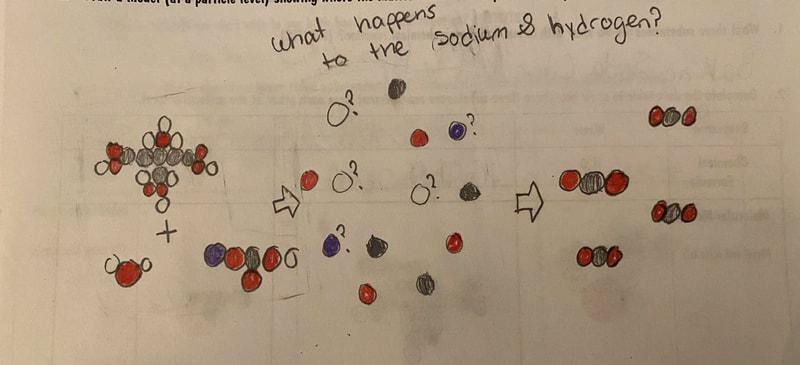
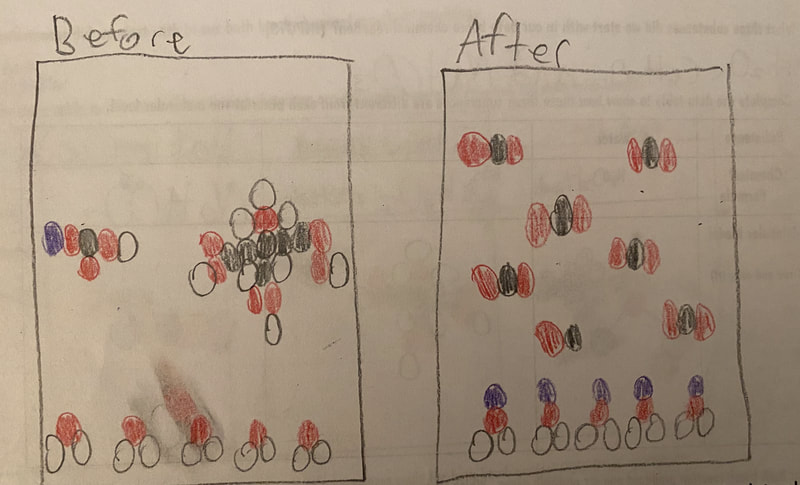
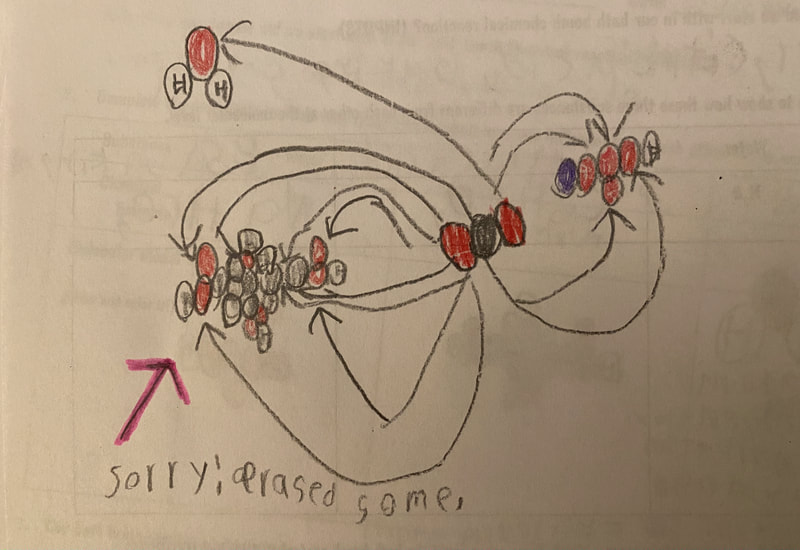
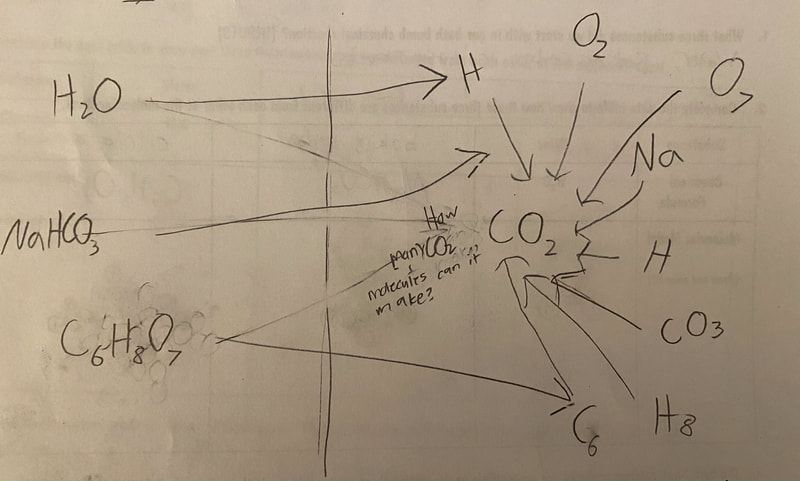
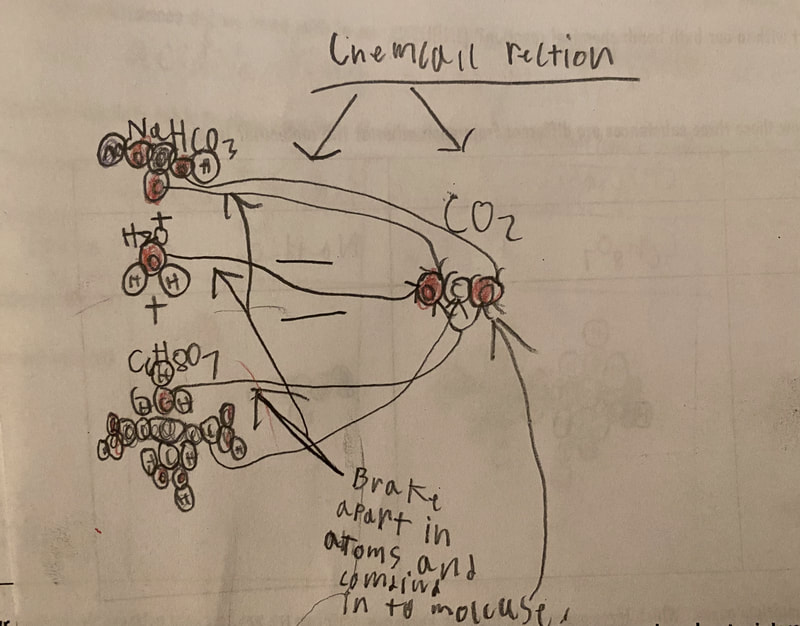
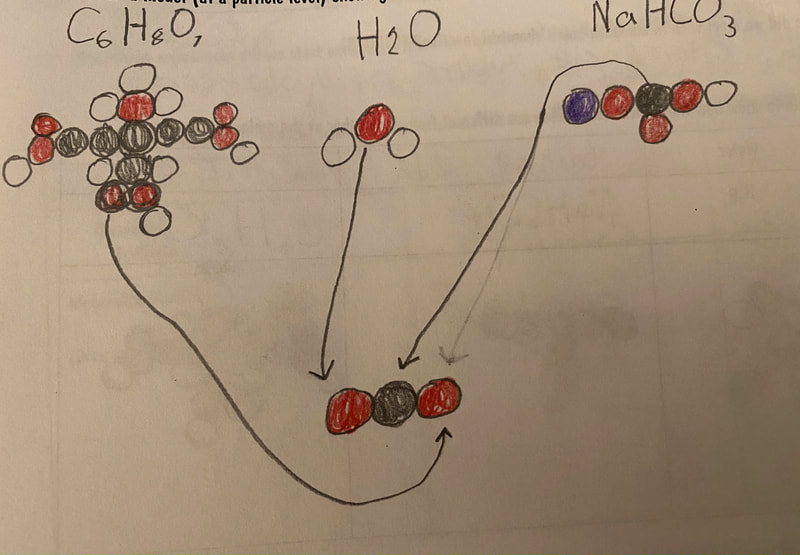
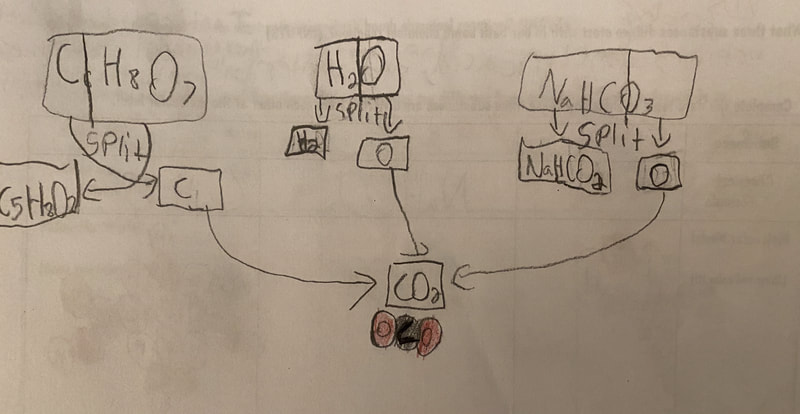
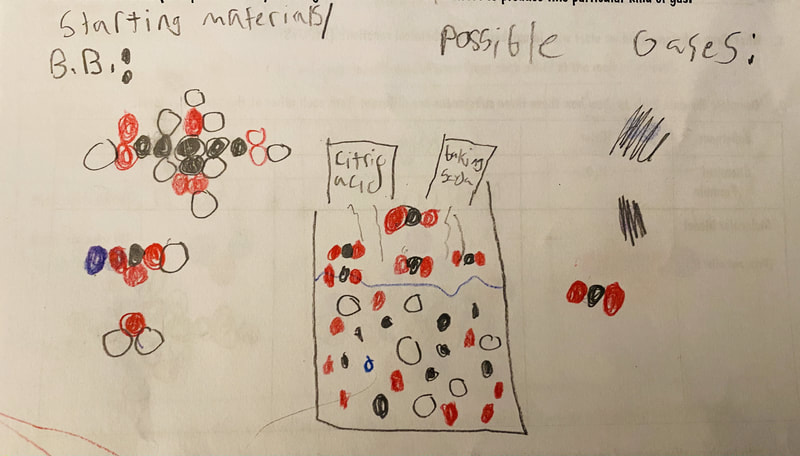
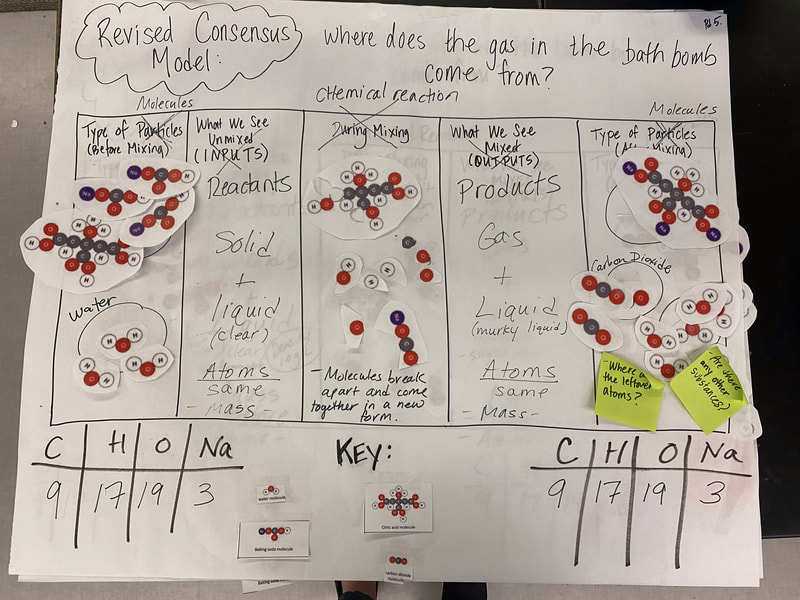
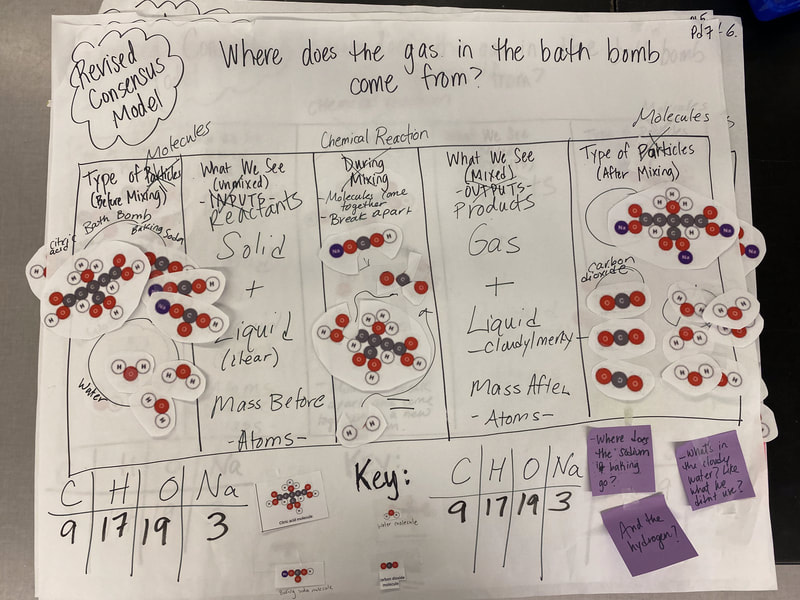
Now that we had evidence to suggest particles really were, we moved forward with representing our ideas no longer with particles, but instead with molecules. We had an awesome time building out various models of water as a start, and then moved towards understanding that different molecules are made of the same atoms but in different amounts, shapes and arrangements. From here, we started to think about how our chemical reaction starts with baking soda, water, and citric acid, and ends up with carbon dioxide! Check out some students' thinking as they tried to model this! And from here, we worked to really push for what a chemical reaction means, and how our reactant side of our chemical reaction must have the same atoms as the product side of our reaction! We've not only identified the mystery bath bomb gas, but we've figured out other products that included water and citric acid! Way to go 6th graders!
|
Mrs. BrinzaSo how exactly doesn't something new get created when it wasn't there before? Archives
March 2022
Categories |





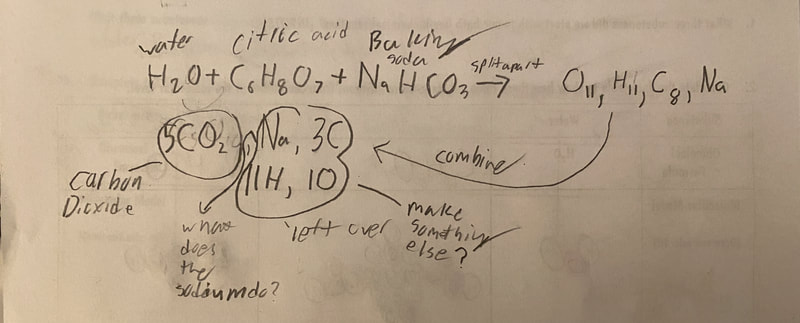

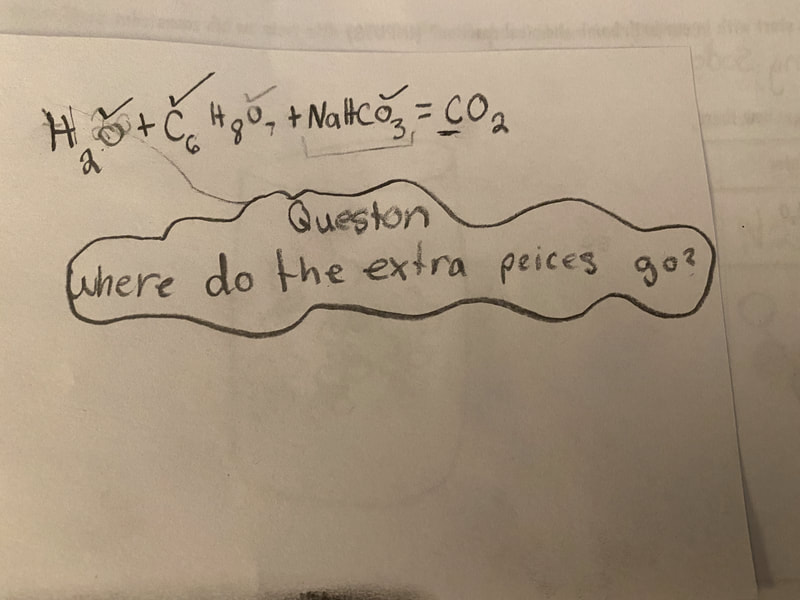

 RSS Feed
RSS Feed