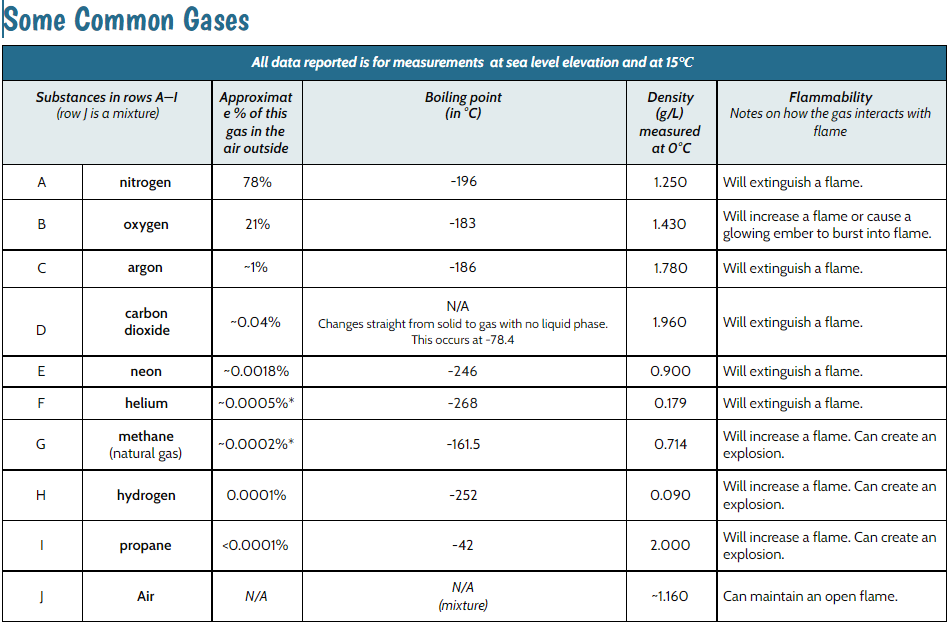
Knowing we could easily get our hands on the air in our classroom and the gas from the bath bomb + water reaction, we figured we could get our hands on some helium, and we did. We examined the properties of various common gases, and created a plan for how we would do various tests on the three gases we had access to. Of course we wanted to do so safely, recognizing that the flammability property could tell us a lot!
So we chose to put a flame to air in our classroom, helium from a party balloon tank, and the gas created from the bath bomb + water reaction. Here's what we found:
| | | |
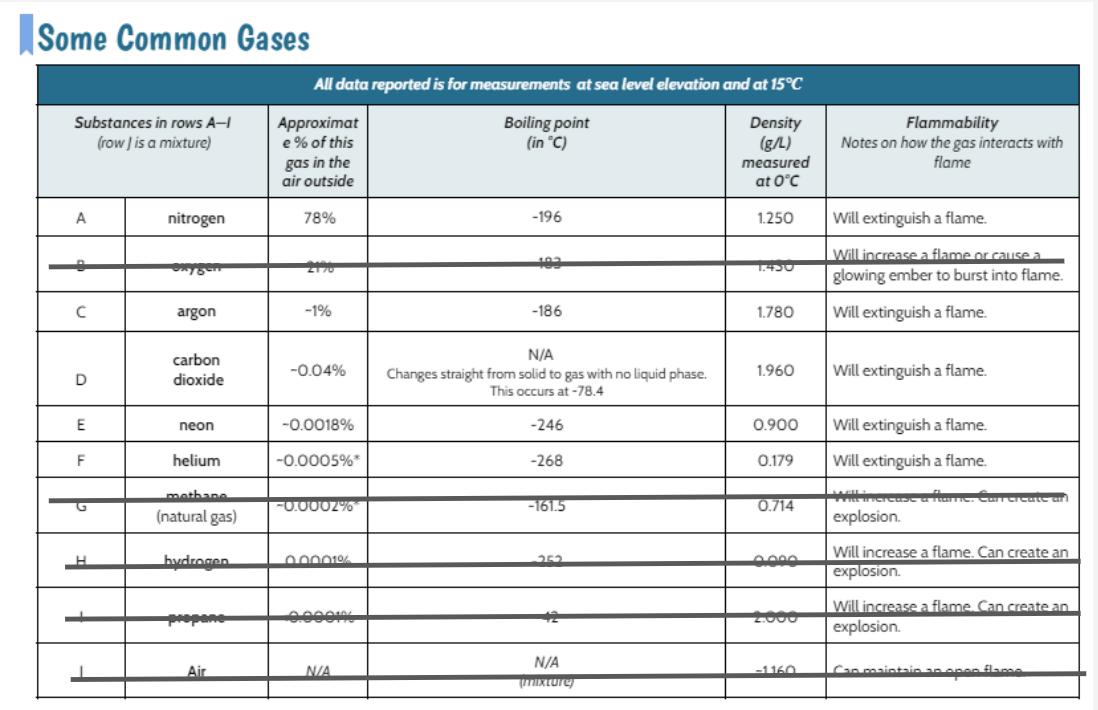
Air was flammable and helium put out the flame (which we knew from our Common Gases Data Table). However, we saw the bath bomb + water gas put out the gas, too! Does this mean it was helium? Looking at our common gases data table, we realized that lots of common gases put out a flame, too, not just helium! It could also be nitrogen, argon, carbon dioxide or neon.
Another test would have to help us! Our freezer at school isn't cold enough for the gases left to condense into water, so maybe exploring density could help alongside flammability.
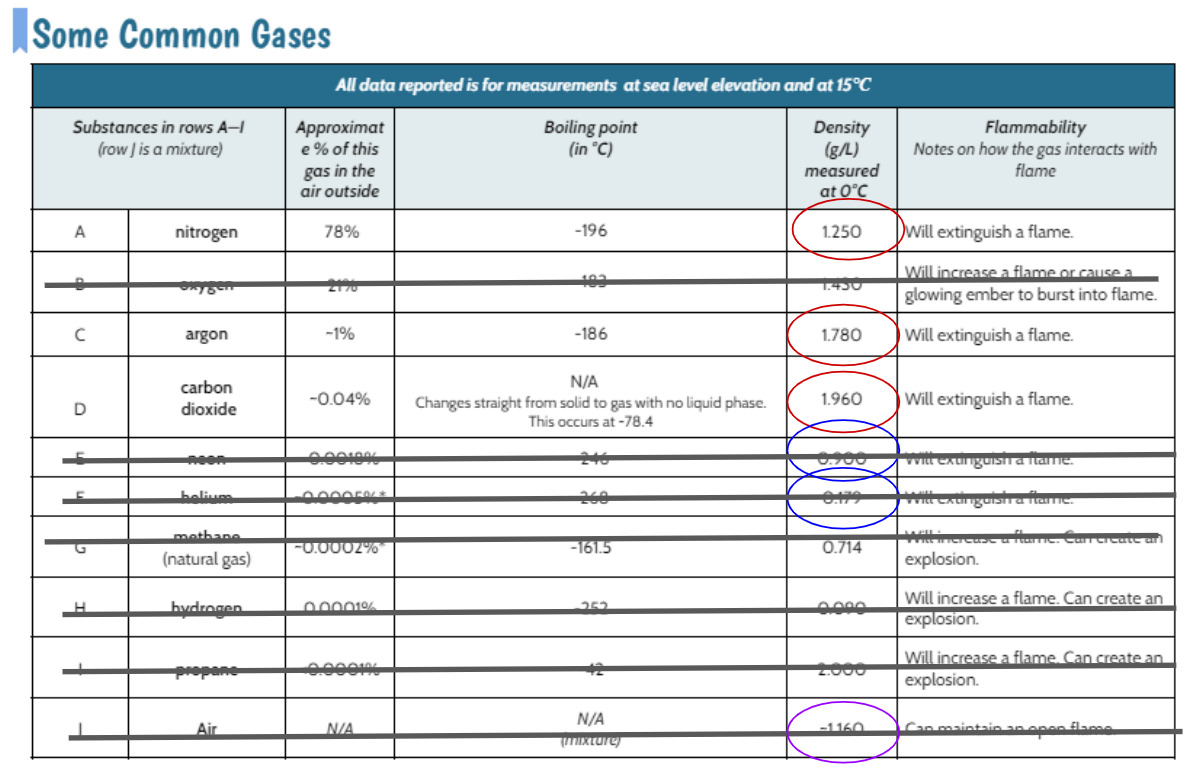
We worked through how changing the location of the flame would help us with the bath bomb gas' density. If the bath bomb gas was less dense than air, it would put out a flame above the gas, yet not below. And if the bath bomb gas was denser than air, it would put out a flame below it but not above.
We worked through how changing the location of the flame would help us with the bath bomb gas' density. If the bath bomb gas was less dense than air, it would put out a flame above the gas, yet not below. And if the bath bomb gas was denser than air, it would put out a flame below it but not above.
| | |
Using the data we gathered from both the flammability and density tests, we could narrow down the bath bomb + water gas to be one of the following:
It could be nitrogen, argon, or carbon dioxide because each of these gases is extinguishes a flame and is less dense than air. How are we going to figure out which gas or gases this reaction is?!?!?!
Looks like we need to review what we've figured out and update our consensus model to see if this helps us move our science thinking forward!
Looks like we need to review what we've figured out and update our consensus model to see if this helps us move our science thinking forward!
 RSS Feed
RSS Feed