Considering the fact that we think what's around the cups changes the cups, we decided we really wanted to think about changing the air temp around the cups. This seemed like a pretty daunting task since whenever we measured air temperature in our classroom, we got all kinds of differences. We agreed that water surrounding cups (like a bath or hot tub) could give the same feeling like a sauna around a cup!
We agreed to change temperatures inside and outside the cup, and record temperatures both at the start and end of a five-minute period.
We agreed to change temperatures inside and outside the cup, and record temperatures both at the start and end of a five-minute period.
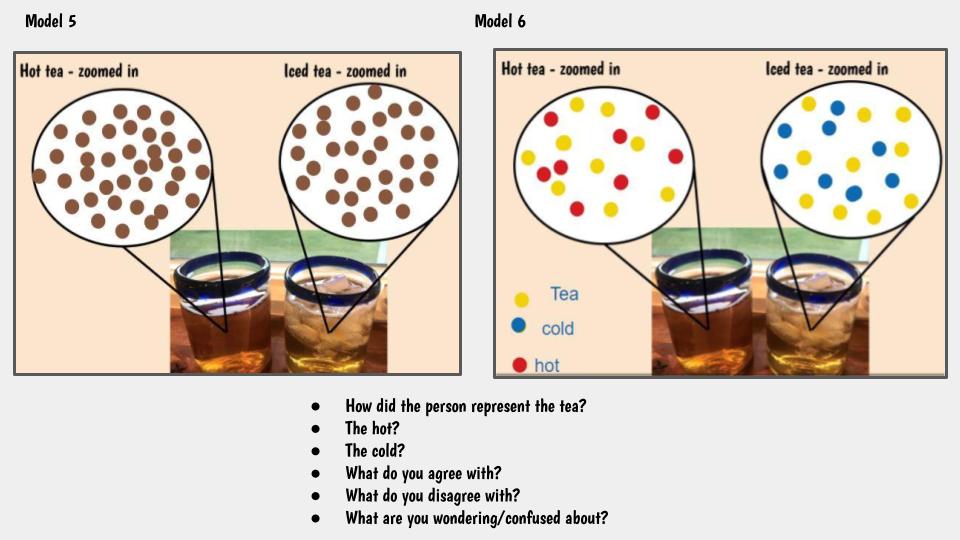
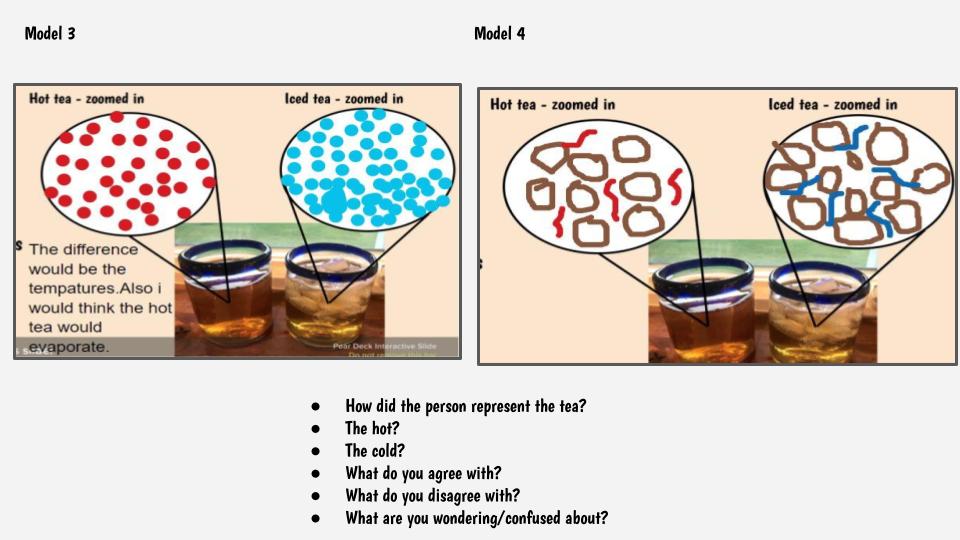
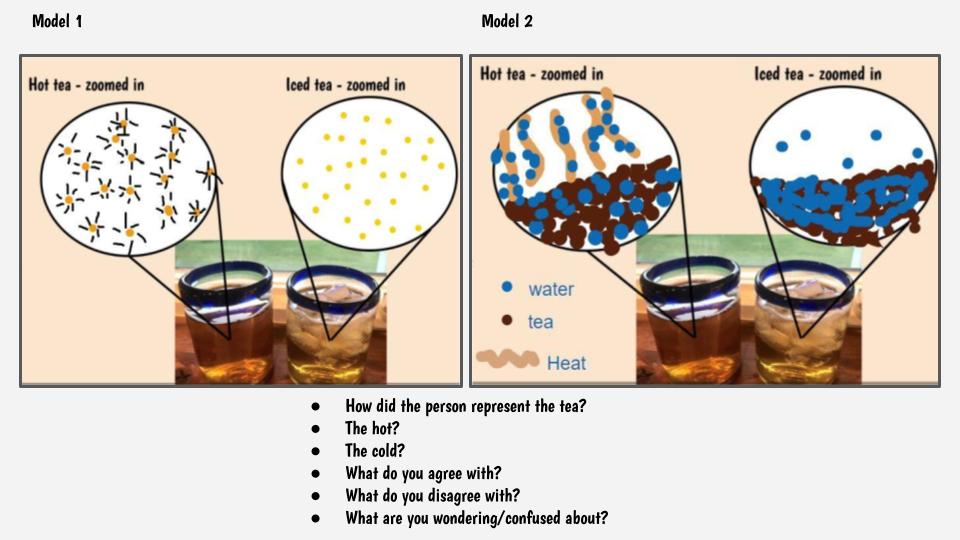
This got us thinking that if hot things always cool down, and cool things always warm up, something has to be getting these changes to happen. We began thinking that there has to be a difference between hot and cold liquids at the particle level, so we considered both hot and cold tea! Students were asked to model the differences and we discussed what we agreed and disagreed with, including areas we still had questions.
We turned to an investigation idea about maybe seeing the differences ourselves, and while just looking at hot, room, and cold water wouldn't cut it, adding food coloring would!
So wait. Our cold cups warmed up. And our warm cups cooled down. Hot liquids move faster than cold liquids. So what's going on with all the particles to show that warmer things have more motion and cooler things have less? Is there a way to actually "see" this, or would a computer based model help?

 RSS Feed
RSS Feed