After designing two rounds of cups, students compared their two designs and made a recommendation as to which cup should be mass-produced! We had some cup designs that didn't change in temperature at all, and some that changed A LOT! We had some expensive designs and others that ran super cheap! We also had some cups that were NOT very environmentally friendly, too! All in all, each of our groups figured out a TON about energy transfer, especially at the particle level! Way to go 6th grade!
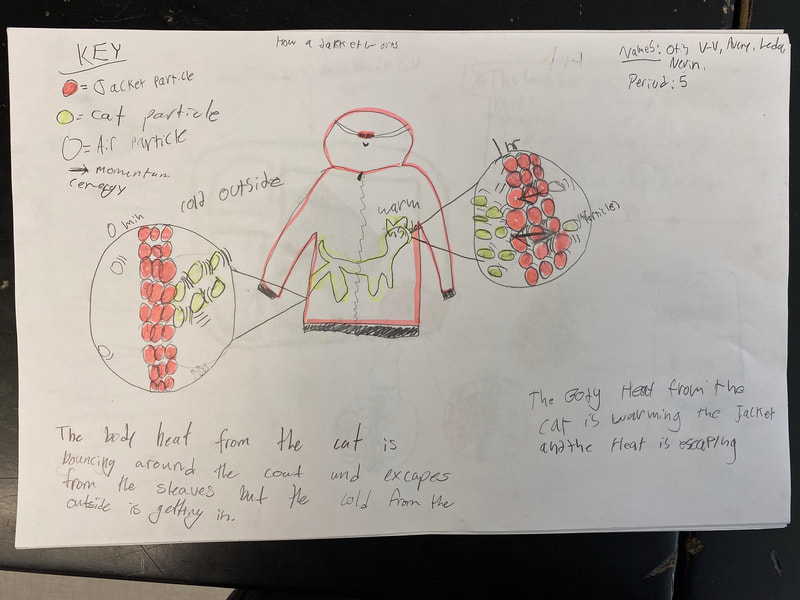
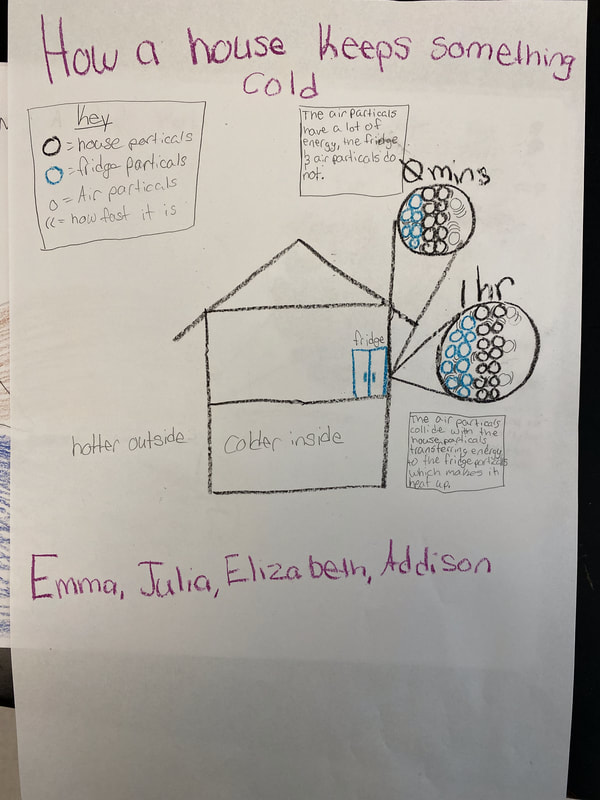
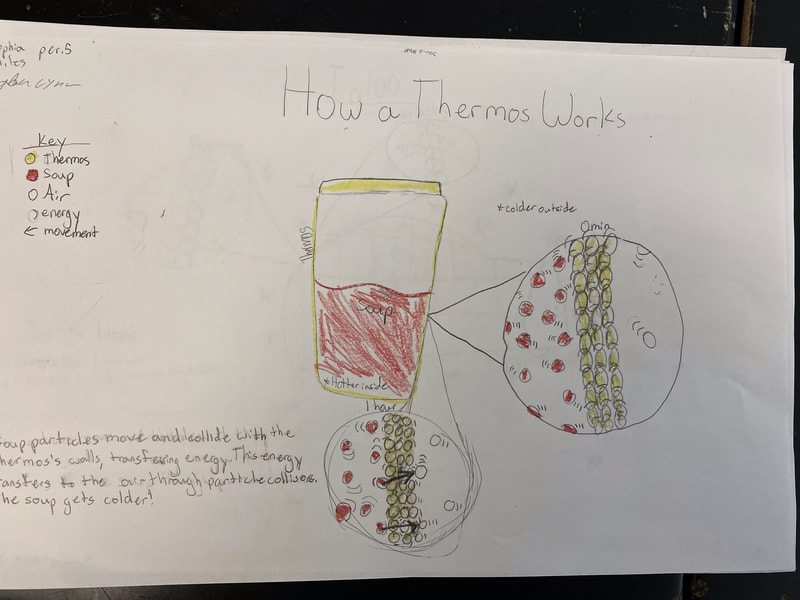
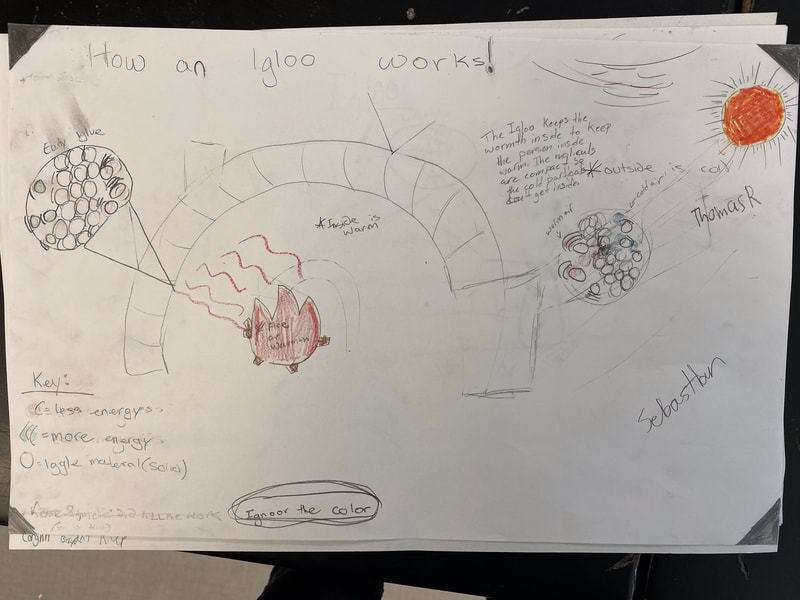
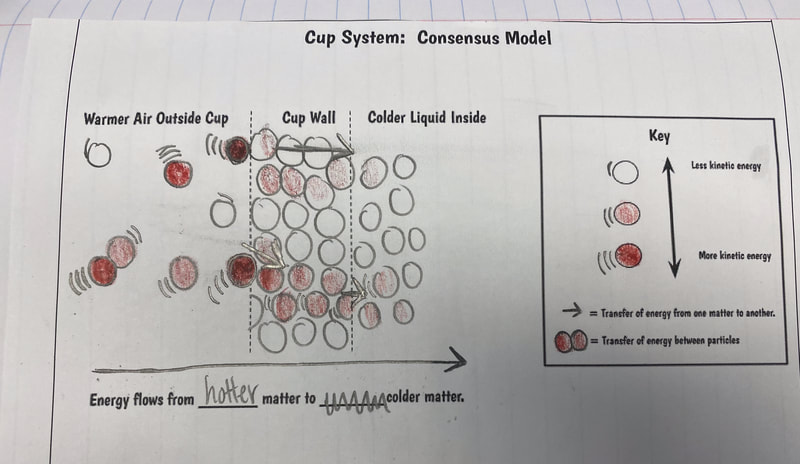
After turning to our related phenomena list from the beginning of the unit, students quickly realized that all the things that they identified that work to keep things hot or cold are just like our cup systems! Working together, we developed a model for another system that each of the classes identified: a cooler! Student groups then picked a related phenomenon and went to town developing a model to explain how it worked to keep something either hot or cold!
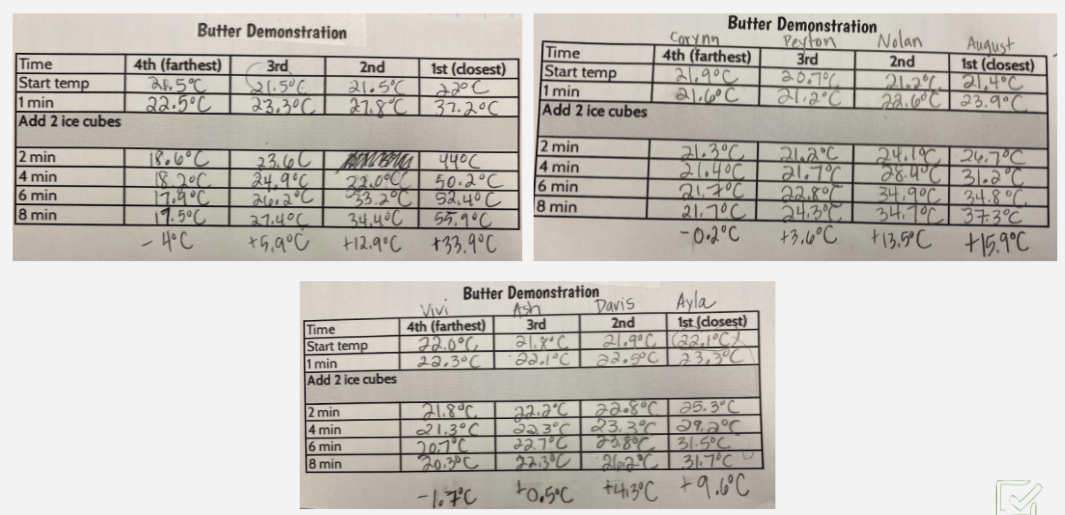
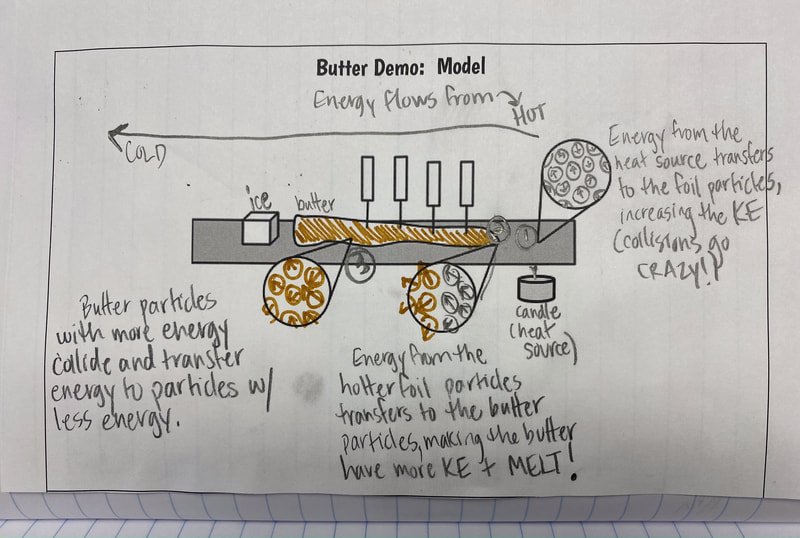
After the long Thanksgiving break, we were ready to jump back in! We reviewed what we figured out about energy transferring between states of matter, and recognized we were still a bit unsure if heat or cold moves. So we turned to something Mrs. Brinza had a surplus of over the holiday...BUTTER! With Mrs. Brinza's guidance, we were thinking that we could see if the temperature changing in butter over time both close to and far from a heat source could give us any solidifying evidence about energy transfer. Check out three class' data below. We agreed that our data helped prove what we were already thinking--that the heat moves. We developed a model to showcase our understanding, and we also considered how what was going on with the butter is just like what's going on with our cup systems! Next steps! Explain our other related phenomena!
|
Mrs. BrinzaSo what is it that keeps things cold or hot? Archives
December 2021
Categories |













 RSS Feed
RSS Feed