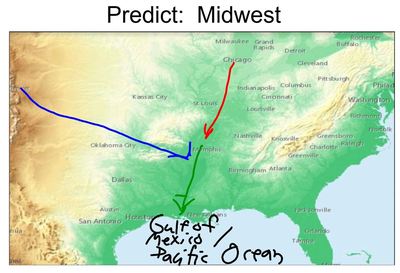
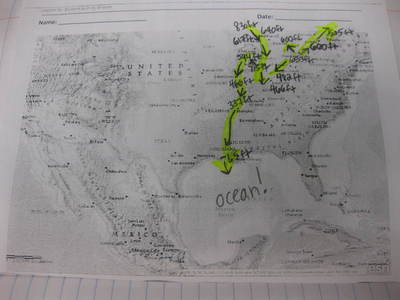
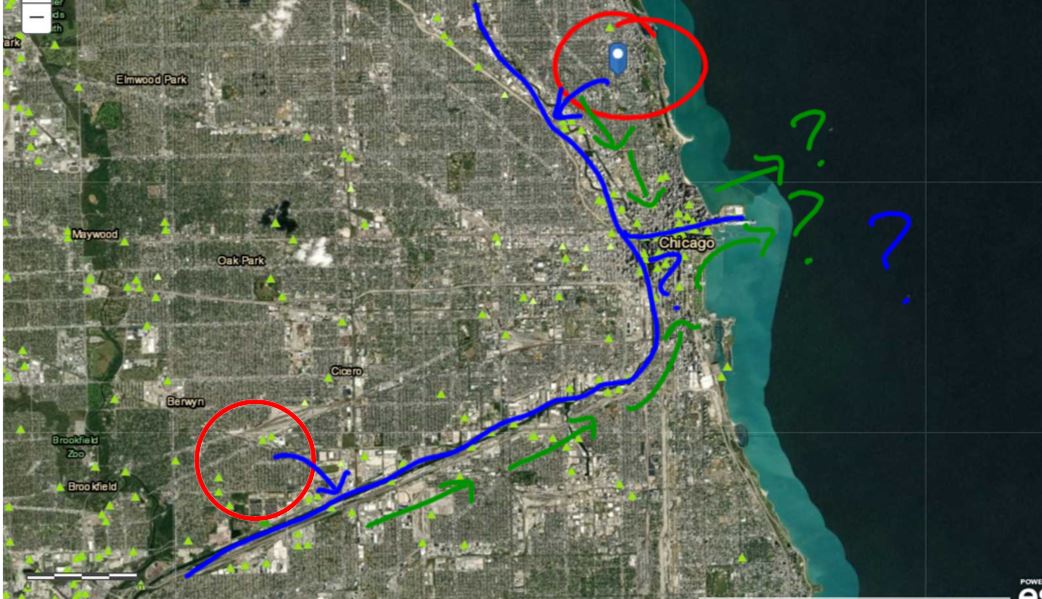
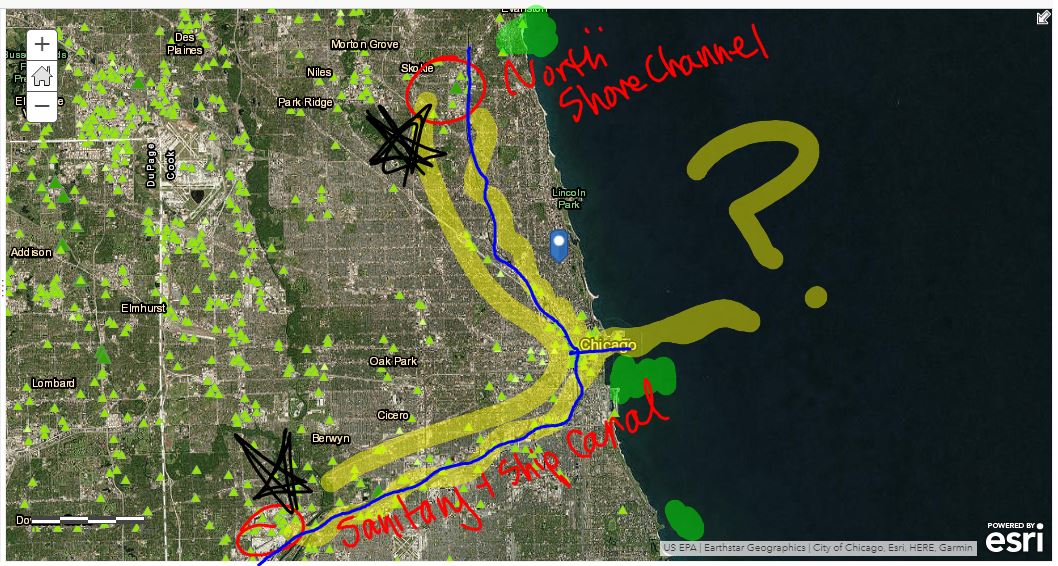
Mrs. Brinza found that data of parts of the city of Chicago were hard to find, but finding elevation data of many cities was fairly easy. So we looked at some maps using color keys that show elevation, along with actual numbers to plot a path the water takes:
We were really interested in where the water goes...and we figured out that all the treated water in Chicago leads to the Chicago River, which flows to the Illinois River, then the Mississippi River. From there, it heads downwards to the Gulf of Mexico, which leads to the Atlantic Ocean.
Yup. The ocean. Which is salt water. This opened up Pandora's box for questioning again.
Yup. The ocean. Which is salt water. This opened up Pandora's box for questioning again.
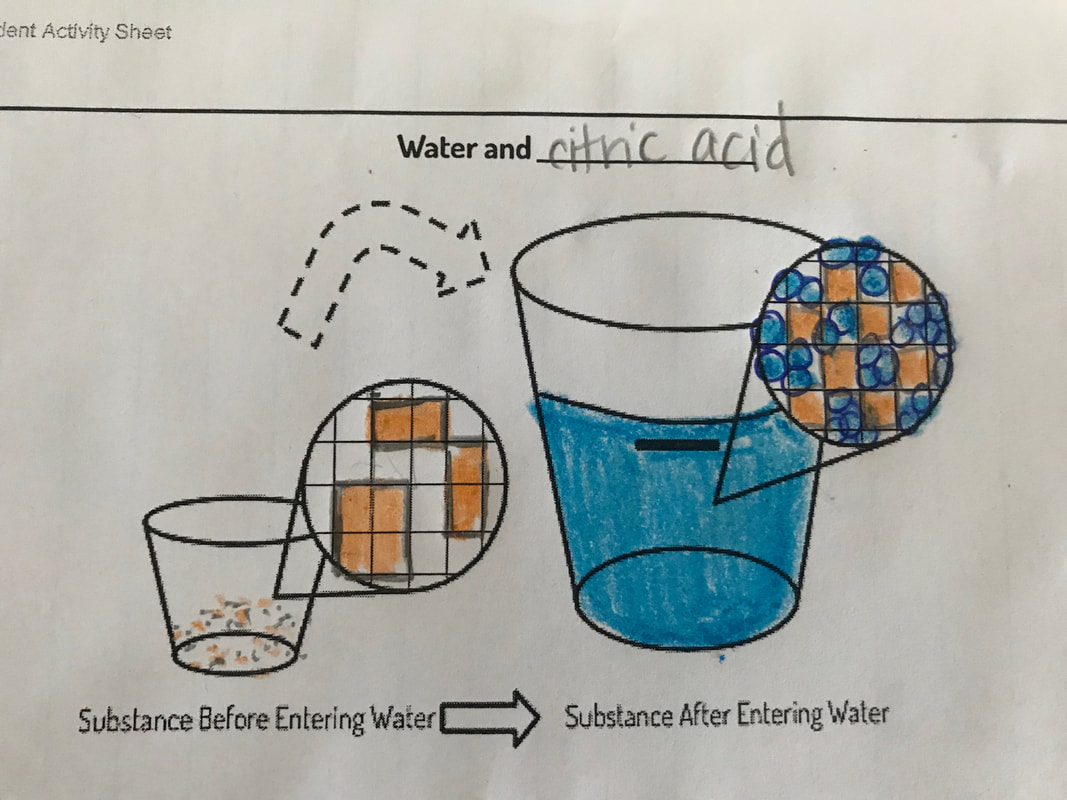
After a lengthy discussion, we agreed to figure out what happens to freshwater when it combines with salt water, find a way to clean salt water to possibly drink it, and then see if this process could be a viable drinking source for the world, since so much of the Earth is covered in salt water. Phew! What a quarter it's been...now we're off to figure out the second part of our Driving Question about our clean drinking water!












































 RSS Feed
RSS Feed