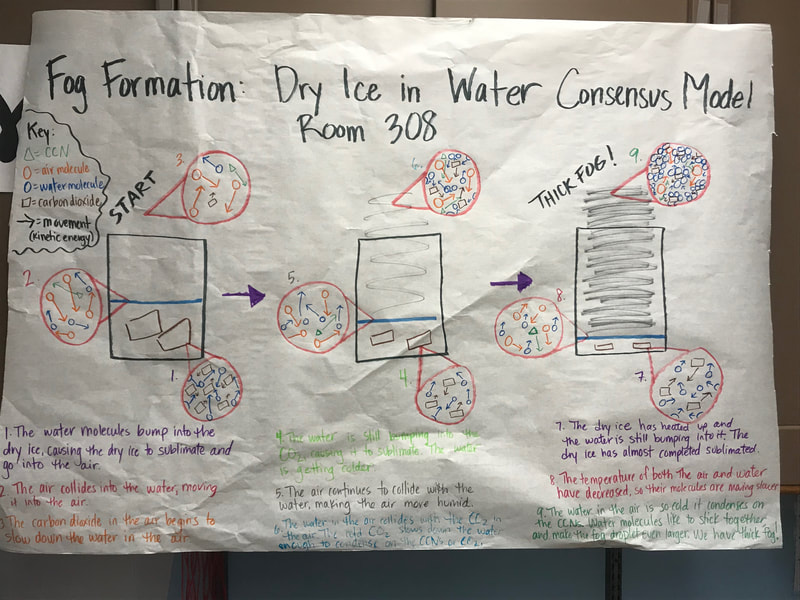
We did it folks! We finally came to consensus today putting together all our science ideas behind what makes fog, and we did it with dry ice and water!
To make a long story short, we figured out the following:
1. When you put dry ice in water, the water molecules have more kinetic energy than the dry ice molecules. This means that when they collide into one another, the energy from the water transfers to the dry ice (solid CO2). This is what gets the dry ice to sublimate (turn from a solid to a gas) and put CO2 into the air.
2. The water in the beaker gets into the air because the air above it is colliding into it, getting it to evaporate. This is making the air more humid. And we know that high humidity is a necessity for fog formation!
3. Once the CO2 and water are in the air, they do two things. First off, we figured out that water molecules are attracted to one another. And since they are still moving, when they bump into the CO2 molecules (which are STILL REALLY, REALLY COLD), they slow down the water molecules enough to get them to condense on either nearby CCNs or the CO2 (which we're thinking is a CCN, too). This high humidity in the air, along with the cooling (because of the dry ice), allows fog droplets to form and fog to appear!
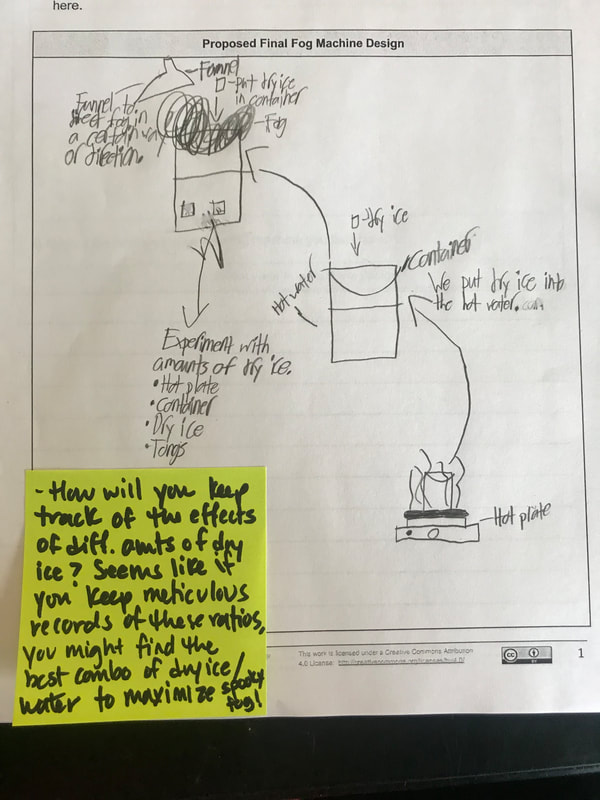
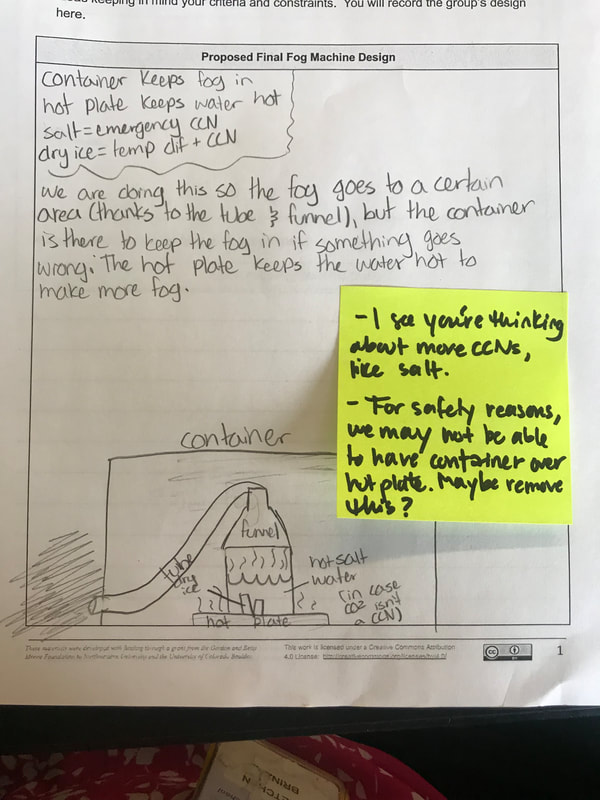
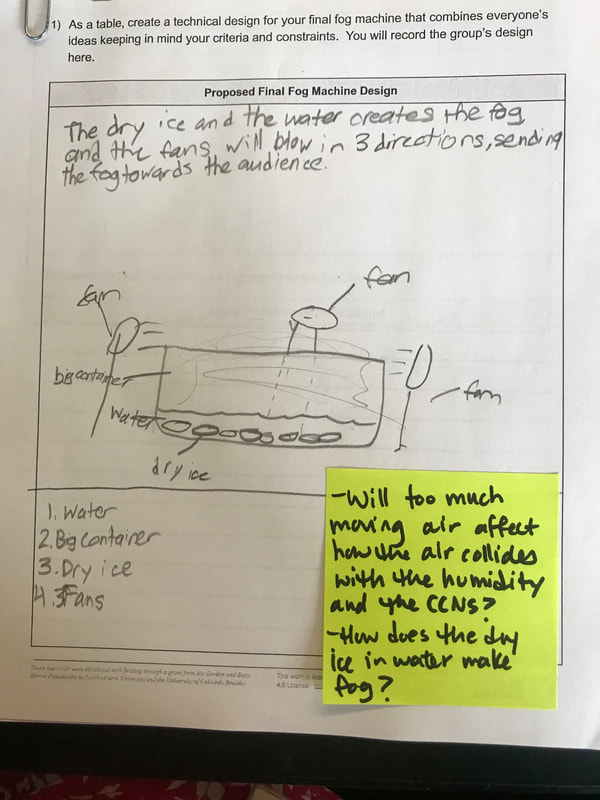
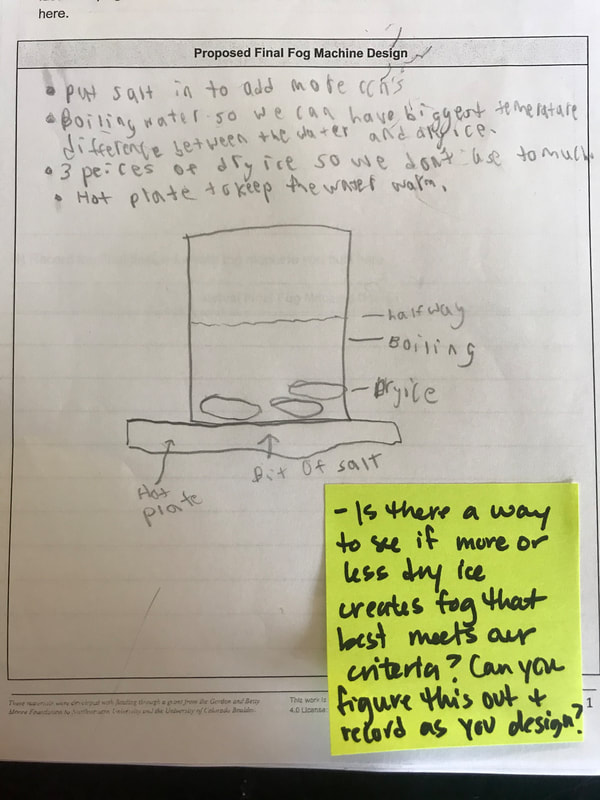
Students are now thinking about their fog machine designs that can best meet the criteria and constraints! Working in teams, they submitted designs to Mrs. Brinza while she secures dry ice. Check out some of the groups' design ideas and some of the feedback Mrs. Brinza gave them.
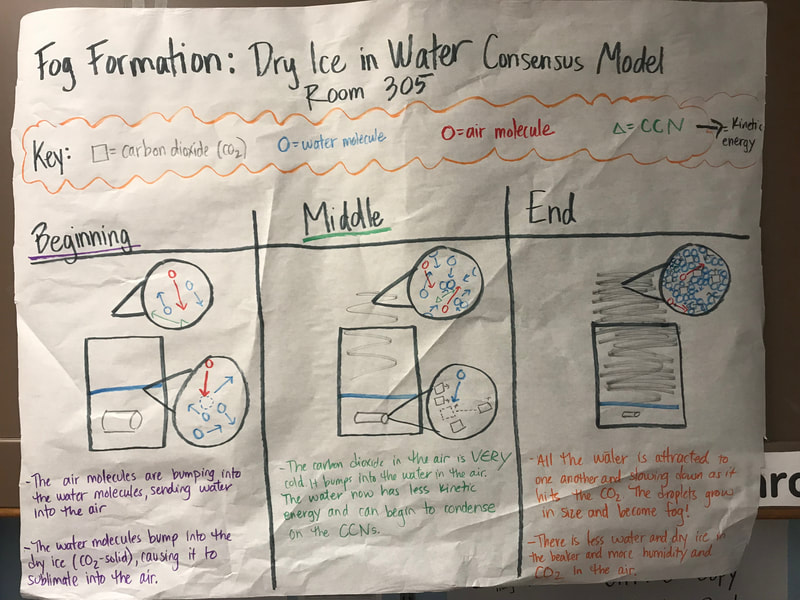
To make a long story short, we figured out the following:
1. When you put dry ice in water, the water molecules have more kinetic energy than the dry ice molecules. This means that when they collide into one another, the energy from the water transfers to the dry ice (solid CO2). This is what gets the dry ice to sublimate (turn from a solid to a gas) and put CO2 into the air.
2. The water in the beaker gets into the air because the air above it is colliding into it, getting it to evaporate. This is making the air more humid. And we know that high humidity is a necessity for fog formation!
3. Once the CO2 and water are in the air, they do two things. First off, we figured out that water molecules are attracted to one another. And since they are still moving, when they bump into the CO2 molecules (which are STILL REALLY, REALLY COLD), they slow down the water molecules enough to get them to condense on either nearby CCNs or the CO2 (which we're thinking is a CCN, too). This high humidity in the air, along with the cooling (because of the dry ice), allows fog droplets to form and fog to appear!
Students are now thinking about their fog machine designs that can best meet the criteria and constraints! Working in teams, they submitted designs to Mrs. Brinza while she secures dry ice. Check out some of the groups' design ideas and some of the feedback Mrs. Brinza gave them.





 RSS Feed
RSS Feed