In our last demonstration, we learned that gas can be added and removed from a container. We saw this as the stopper on top of a flask was sucked in and pushed off! Because gas is made of matter and has mass, the mass of the flask increased as gas was added and decreased as gas was released.
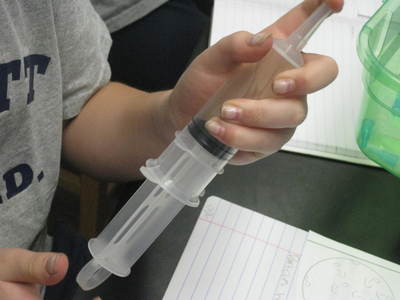
But today's lab involved students keeping the mass constant and changing the volume. Increasing the volume of the syringe allowed students to see that air can expand and spread out. The mass didn't change because no air was added to the syringe to therefore change its mass. When the volume of the syringe decreased, the air inside was compressed, and moved into a smaller space. The mass also remained constant because no more air was added or removed from the syringe.
We'll slowly discover how the ideas behind air's ability to expand and compress relates to why we can possibly smell an odor from a distance. Check back soon!
But today's lab involved students keeping the mass constant and changing the volume. Increasing the volume of the syringe allowed students to see that air can expand and spread out. The mass didn't change because no air was added to the syringe to therefore change its mass. When the volume of the syringe decreased, the air inside was compressed, and moved into a smaller space. The mass also remained constant because no more air was added or removed from the syringe.
We'll slowly discover how the ideas behind air's ability to expand and compress relates to why we can possibly smell an odor from a distance. Check back soon!

 RSS Feed
RSS Feed