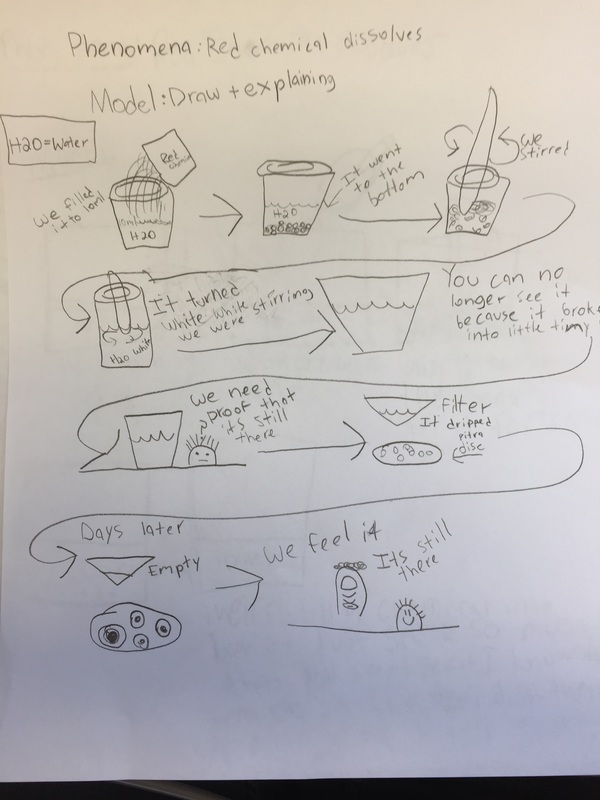
| The results are in! Some of our mystery chemicals dissolved, and this means they were able to be broken down by the water into small enough pieces that they could pass through our filter into the dish. After letting the water evaporate overnight, there was evidence that some of the chemicals did indeed get small enough to pass through the filter! Those chemicals that remained behind in the filter were unable to be broken down by the filter and didn't dissolve. So what does this mean? Not all chemicals that are white and powdery react to water in the same way! How cool! |
We added more questions to our Driving Question Board and agreed that the next best investigation would be to add more than the 6 drops. We agreed to 10 mL!
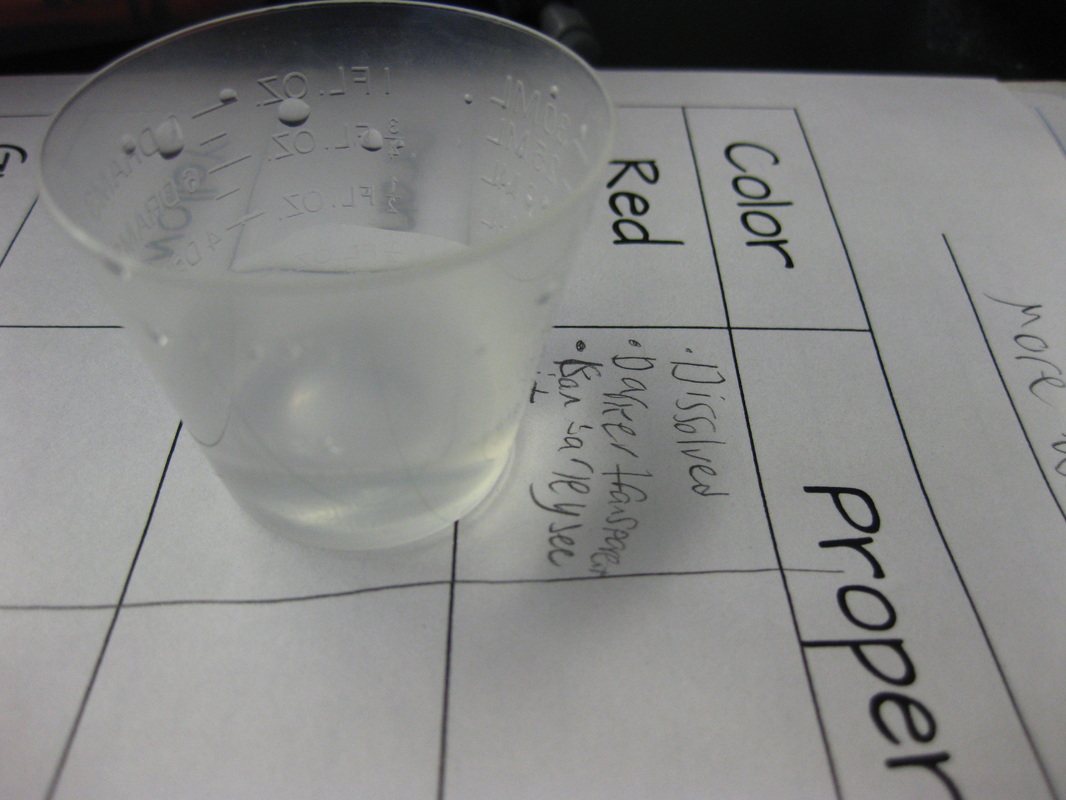
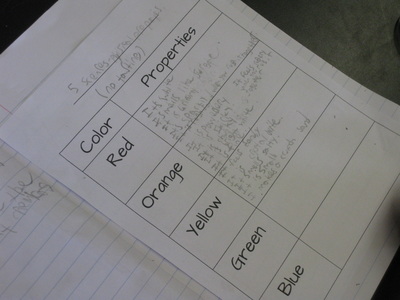
We're discovering all kinds of interesting phenonmena...and are seeing how these events can help identify each of the mystery chemicals. Our first test revealed that the red chemical dissolved. But what does it mean for a chemical to dissolve? We'll be working on the practice of modeling to explain this! After learning about our mystery chemical's physical and chemical properties, we're digging deeper into our five mystery chemicals. Each of the chemicals has its own unique properties, yet they all share one physical property in common--they're white!
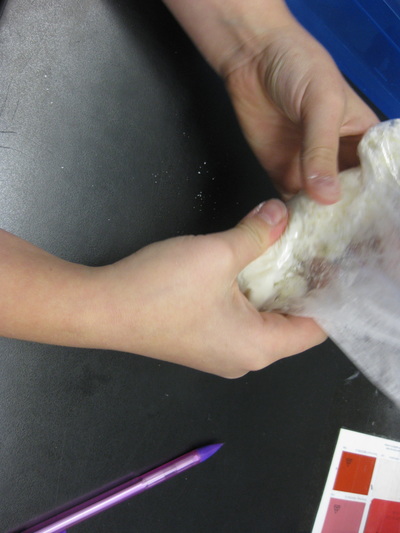
Just how can we tell the difference between them? We looked at texture, noise, and odor to name a few. Our next steps will be to see if there are any unique chemical properties between them! So as fifth graders dig deeper into understanding the basics of chemistry, they are discovering that not all chemicals have the same properties. Taking our mystery chemical, students decided to add water to it to see how it reacted. The results surprised them for sure, discovering new properties about the substance!
We'll be using the ideas behind developing investigations and analyzing data to help us state claims about what each mystery chemical is. It's certainly exciting in our classroom! We kicked off our Chemical Tests Unit by working with a mystery chemical. Learning about physical and chemical properties, students are recognizing that each chemical has unique properties that help with its identification. Over the course of the unit, students will develop investigations and analyze their findings to identify five mystery chemicals.
They'll then communicate their findings using the Claim-Evidence-Reasoning model we've been working on since the beginning of the year. Fifth graders are certainly walking the walk and talking the talk of scientists! |
Mrs. BrinzaIt's amazing to see when students use science and math in ways in which correlate to class! Keep up the great work 5th grade! Archives
December 2016
Categories |










 RSS Feed
RSS Feed