Here's what we've seen so far!
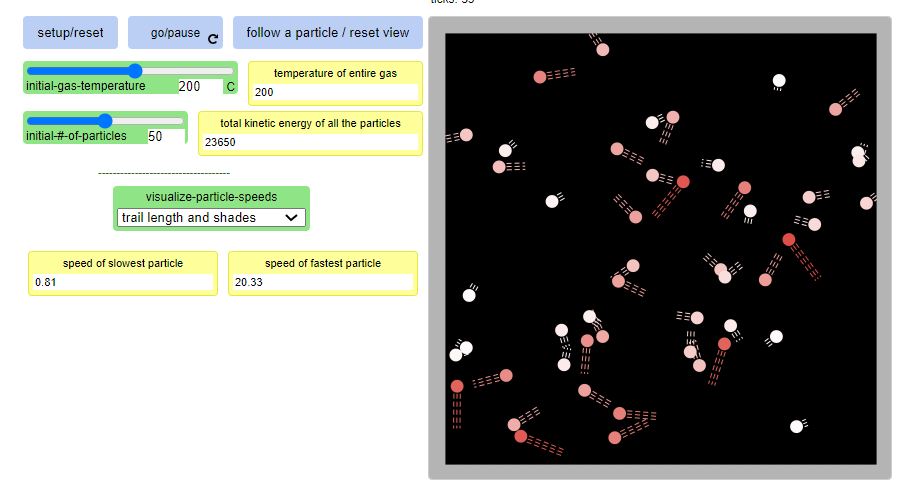
1. Increasing the initial temperature of the gas makes the particles move faster.
2. All the particles in the gas don't move at the same speed...some move really fast and others more really slowly!
3. When a fast particle hits a slower particle, the fast particle slows down and the slow particle speeds up (kind of like in a car crash with a parked car).
4. Even though particles are constantly speeding up and slowing down, the overall temperature stays the same.
5. When you add more particles and keep the temperature the same, we see an increase in energy. This is making sense to us as every particle has energy...so more particles means more energy!
We continue to use the simulator to help us figure out what's going on with our cups to make our hot drinks cool down and our cold drinks warm up!
1. Increasing the initial temperature of the gas makes the particles move faster.
2. All the particles in the gas don't move at the same speed...some move really fast and others more really slowly!
3. When a fast particle hits a slower particle, the fast particle slows down and the slow particle speeds up (kind of like in a car crash with a parked car).
4. Even though particles are constantly speeding up and slowing down, the overall temperature stays the same.
5. When you add more particles and keep the temperature the same, we see an increase in energy. This is making sense to us as every particle has energy...so more particles means more energy!
We continue to use the simulator to help us figure out what's going on with our cups to make our hot drinks cool down and our cold drinks warm up!
 RSS Feed
RSS Feed