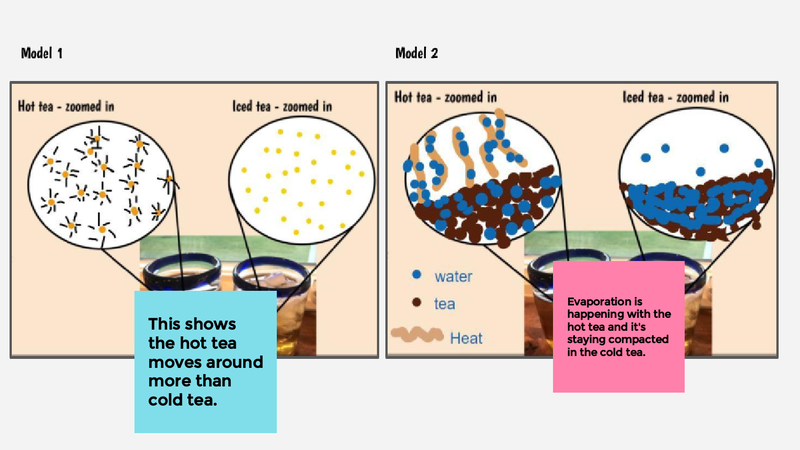
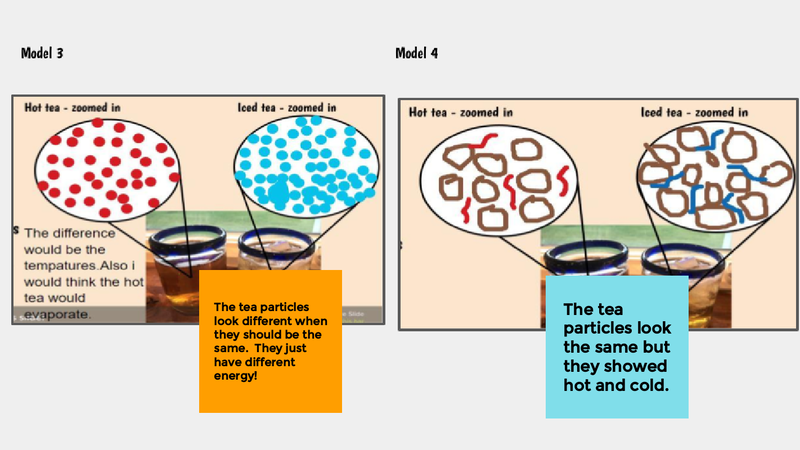
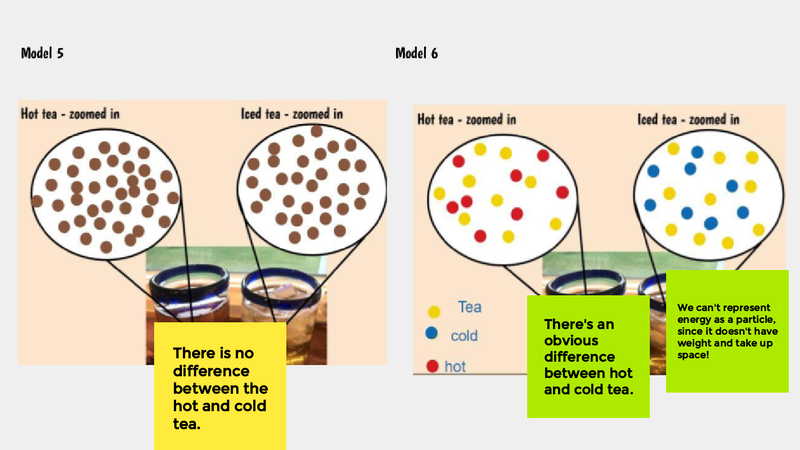
Recognizing a bunch of patterns from our water bath experiments, we are recognizing the power of how a model can help us understand what is going on with both both hot and cold water when they are near each other.
In order to do this, we first have to figure out what hot things and cold things actually look like on the molecular level, so we had some class discussions about each of the models below.
In order to do this, we first have to figure out what hot things and cold things actually look like on the molecular level, so we had some class discussions about each of the models below.
Thinking about what hot and cold things "do" really got us thinking. We know that hot things cool down over time, and that cold things warm up, too. So we're trying to think about what the particles/molecules are really up to. How is it that they get more or less energy over time? We turned to YouTube for some guidance to consider what happens when we begin to "SEE" what's going on with things that really are oh so tiny!
We saw that the candy in the hot liquid began to melt/dissolve/remove the red coloring faster than when the candy was placed in the cold water. But it was so interesting to see that even in the cold water, the red part of the candy came off. We know that the only way something can come off of something else is with force. So is the water in the cups really moving? And knocking on the candy? We really wished we could zoom in on the water to see what it was doing!
 RSS Feed
RSS Feed