Our two dimensional models and ball-and-stick models do not show how the molecules in a substance are in constant motion. But a computer simulation will!
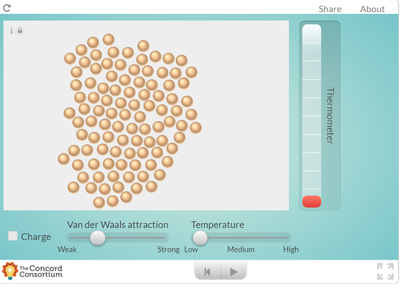
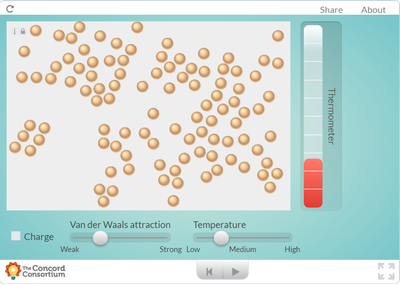
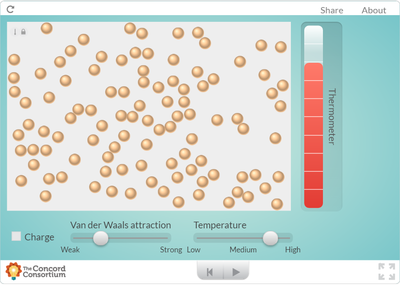
Using an online program, students saw what happens to molecules as they are heated. How does heating a substance affect its motion? How does it affect the volume of a substance? Take a look for yourself...
But it seems like when there is more energy in a substance, the molecules move faster and take up more space. Is this why I smell a hot pizza quicker across a room than a cold pizza? Hmmmmm....
Using an online program, students saw what happens to molecules as they are heated. How does heating a substance affect its motion? How does it affect the volume of a substance? Take a look for yourself...
But it seems like when there is more energy in a substance, the molecules move faster and take up more space. Is this why I smell a hot pizza quicker across a room than a cold pizza? Hmmmmm....



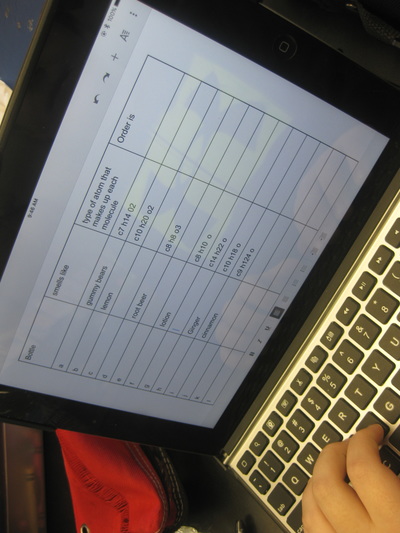

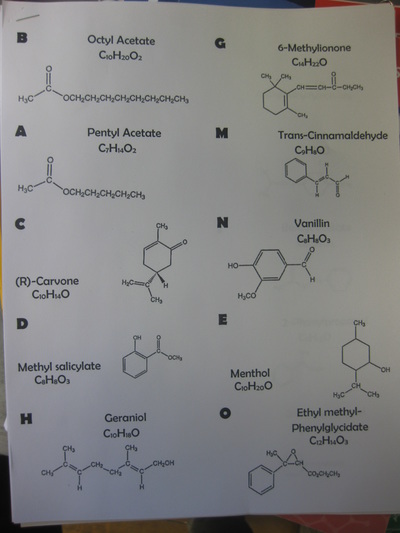
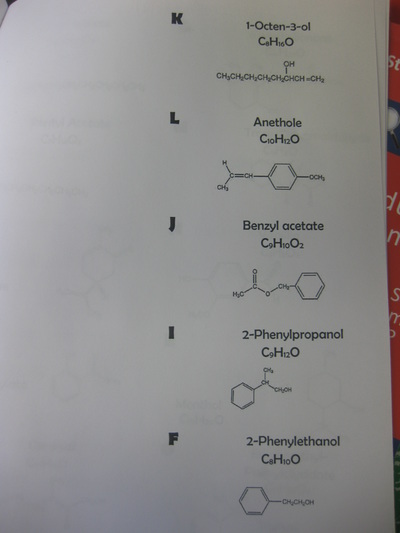
 RSS Feed
RSS Feed